Simplify Life®
with Omnipod® 5

#1 Prescribed Automated Insulin Delivery System1
Omnipod 5 provides automated insulin delivery for up to 3 days (72 hours) without daily injections.
The system proactively helps to correct highs, protect from lows, and has been shown to lower A1c2-4 so you can spend more time living life and less time thinking about diabetes.
Get to know the Omnipod 5 System
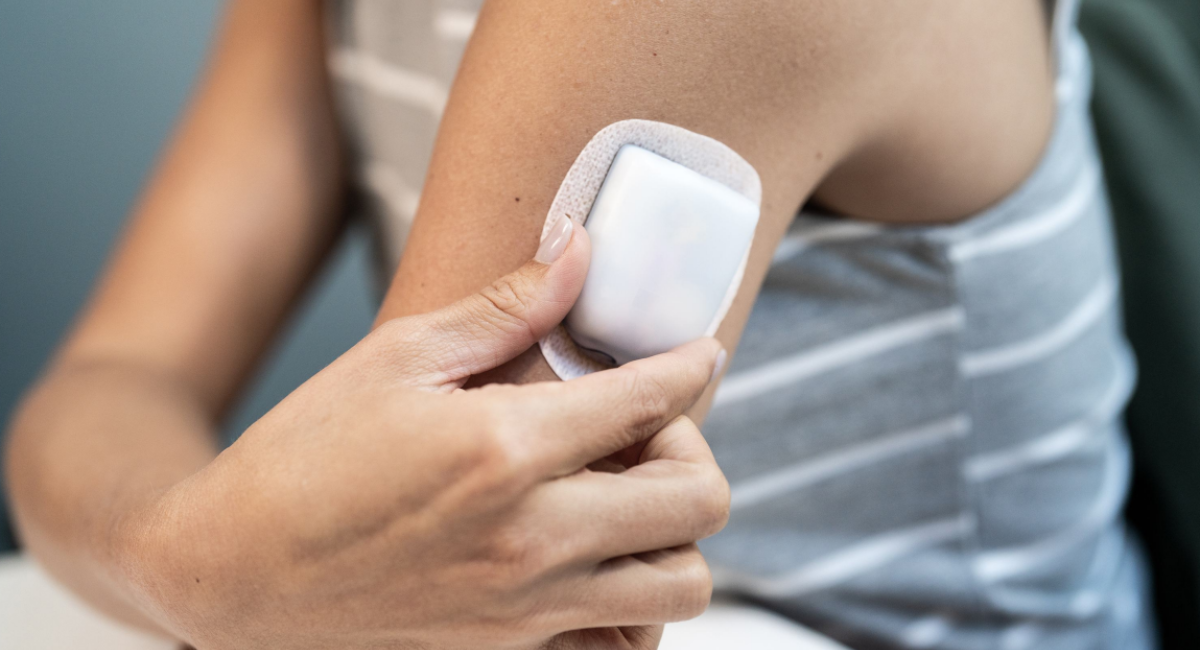

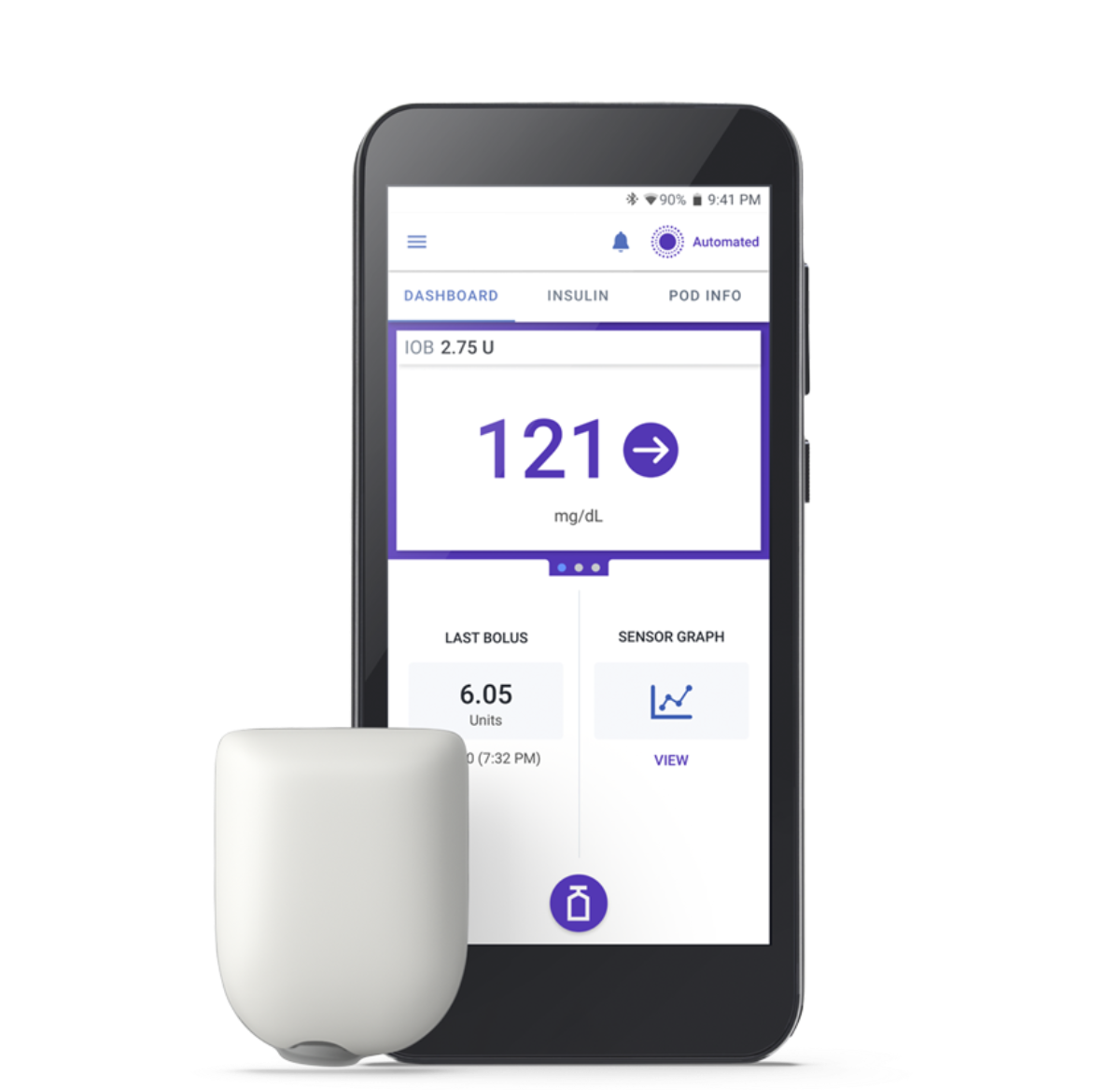
The Pod
Wearable and waterproof**—sits right on your body
- Easy to apply: fill the Pod with up to 200 units of U-100 rapid acting insulin and place it almost anywhere you would give an injection
- The small flexible cannula inserts automatically, you'll never see it and barely feel it
- Small enough to be worn under clothing


The Omnipod 5 App
Makes insulin delivery look like sending a text
- Use a compatible smartphone† or the Insulet provided Controller to control the App
- Includes features like SmartBolus Calculator, Activity feature, and Custom Foods
- Displays glucose and insulin information so you can easily access your data
How it works
The Omnipod 5 algorithm is built right into the Pod. It's the brains behind the system helping keep you in range day and night.5-7 Here's how:
- Predicts future glucose levels up to 60 minutes ahead using your CGM sensor value and trend.
- Adjusts insulin delivery based on a customizable target, ranging from 110 mg/dL-150 mg/dL.
- Automatically increases, decreases or pauses insulin every 5 minutes.
Bolus doses can be given for meals using the Omnipod 5 App or Controller.
Get the full experience with a CGM sensor
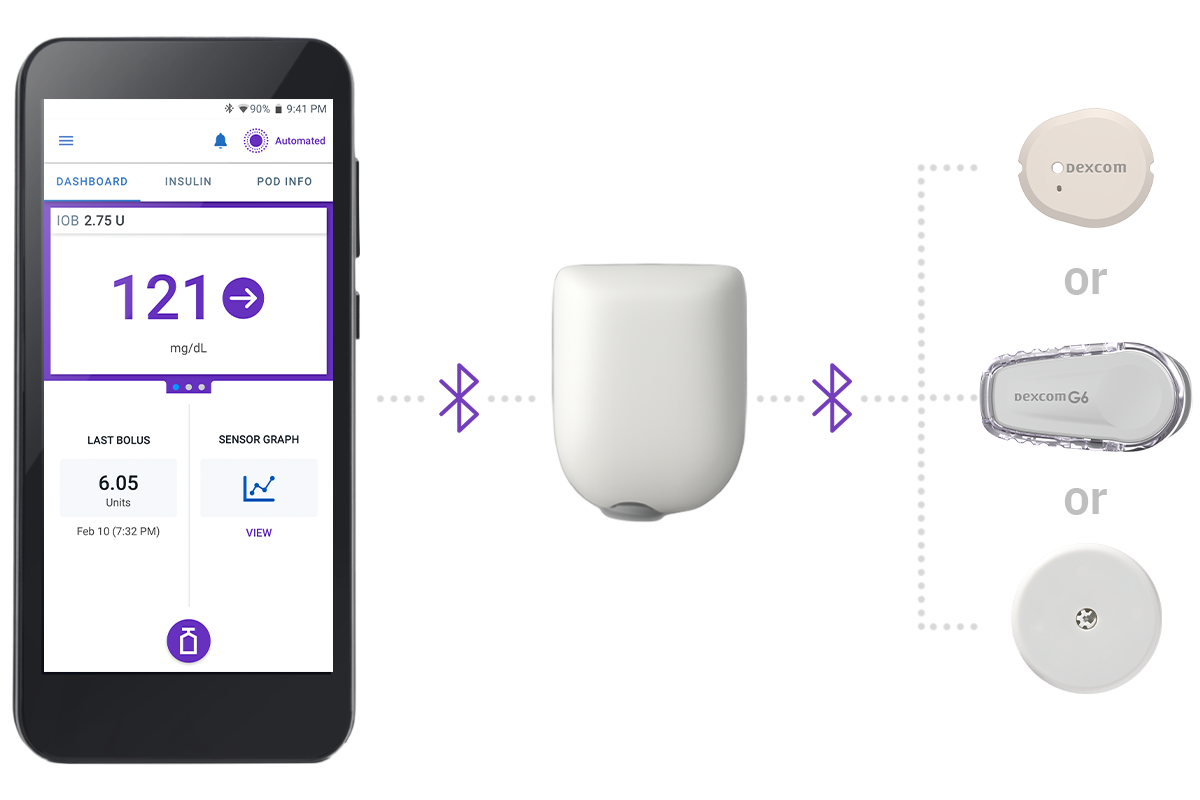

In continuous communication, your Continuous Glucose Monitor (CGM) sensor sends glucose values and trends to the Pod to help the system make automated insulin delivery decisions. Omnipod 5 is compatible with Dexcom G7 CGM Systems, Dexcom G6 and FreeStyle Libre 2 Plus Sensor.
Dexcom is in the process of discontinuing Dexcom G6 to focus on delivering the next generation of CGM. Omnipod 5 will continue to support integration with Dexcom G6 for as long as this sensor remains available. For more information, please visit www.dexcom.com or contact Dexcom support.
Real people, real stories
“Having control over my diabetes and my numbers—I’ve found this new confidence that I didn’t have before.”
Morgan M.
Omnipod 5 User
Simple ways to get started
Omnipod 5 is available through the pharmacy, which means you can make the switch at almost any time.‡
It all starts with a free, no obligation, benefits check.
I'd like to check my coverage
Find out your estimated copay
I'd like a free trial of Omnipod§
Take the Pod for a test drive
Looking for more information?
Learn more about Omnipod 5 for type 1 diabetes or type 2 diabetes or check out our library of resources including:


**The Pod has an IP28 rating for up to 25 feet for 60 minutes. The Controller is not waterproof.
†For a list of compatible smartphone devices visit omnipod.com/compatibility.
‡Depending on individual insurance coverage, limitations and approvals.
1. USA 2024, Data on file,
2. Brown S. et al. Diabetes Care (2021). Study in 240 people with T1D aged 6 - 70 years involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Average time with high blood glucose in adults/adolescents and children, standard therapy vs 3-month Omnipod 5: 32.4% vs. 24.7%; 45.3% vs. 30.2%. Average time with low blood glucose in adults/adolescents and children, standard therapy vs 3-month Omnipod 5: 2.9% vs. 1.3%; 2.2% vs. 1.8%. Average A1c in adults/adolescents and children, standard therapy vs. Omnipod 5 = 7.16% vs 6.78%; 7.67% vs 6.99%. Study funded by Insulet.
3. Sherr JL, et al. Diabetes Care (2022). Study in 80 people with T1D aged 2 - 5.9 yrs involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Average time with high blood glucose in very young children, standard therapy vs 3-month Omnipod 5: 39.4% vs. 29.5%. Average time with low blood glucose in very young children, standard therapy vs 3-month Omnipod 5: 3.43% vs. 2.46%. Average A1c in standard therapy vs. Omnipod 5 = 7.4% vs. 6.9%. Study funded by Insulet.
4. Pasquel FJ, et al. Presented at: ADA; June 21-24, 2024; Orlando, FL. Prospective pivotal trial in 305 participants with T2D aged 18-75 yrs. Study included a 14-day standard therapy (ST) phase followed by a 13-week Omnipod 5 hybrid closed-loop phase. Mean time >180 mg/dL as measured by CGM: ST = 54%, 3-mo Omnipod 5 = 34%, P<0.001. Mean time <70 mg/dL as measured by CGM: ST = 0.2%, 3-mo Omnipod 5 = 0.2%. Mean HbA1c: ST vs. 13-week Omnipod 5: 8.2% vs. 7.4%, P<0.001.
5. Brown et al. Diabetes Care (2021). Study in 240 people with T1D aged 6 - 70 years involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Average time (6AM-12AM) in Target Glucose range (from CGM) in adults/adolescents and children for standard therapy vs Omnipod 5 = 64.8% vs. 72.5%; 51.5% vs. 64.6%. Average time (12AM-6AM) in Target Glucose range (from CGM) in adults/adolescents and children for standard therapy vs Omnipod 5 = 64.3% vs. 78.1%; 55.3% vs. 78.1%.
6. Sherr JL, et al. Diabetes Care (2022). Study in 80 people with T1D aged 2 - 5.9 yrs involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Average time in Target Glucose range (6AM-12AM) from CGM in standard therapy vs Omnipod 5 = 56.9% vs. 63.7%. Average time in Target Glucose range (12AM-6AM) from CGM in standard therapy vs. Omnipod 5 = 58.2% vs 81.0%.
7. Pasquel FJ, et al. JAMA Network Open (2025). Prospective pivotal trial in 305 participants with T2D aged 18-75 yrs. Study included a 14-day standard therapy (ST) phase followed by a 13-week Omnipod 5 hybrid closed-loop phase. Mean overnight time 70-180 mg/dL (12AM-6AM) as measured by CGM: ST = 49%, 3-mo Omnipod 5 = 70%. Mean daytime 70-180 mg/dL (6AM-12AM) as measured by CGM: ST = 44%, 3-mo Omnipod 5 = 64%. Statistical testing not done to assess significance of change between ST phase and Omnipod 5 System phase.
§Omnipod® 5 Intro Kit 30-Day Trial
1. Program Eligibility
Eligibility criteria: Subject to program limitations and terms and conditions, the Omnipod 5 Intro Kit 30-day trial program (the “Program”) is open to patients who have a valid Omnipod 5 prescription as well as a compatible CGM prescription and who have commercial or private insurance, including plans available through state and federal healthcare exchanges. In order to be eligible, the patient’s eligible insurance plan must include coverage for Omnipod 5 Pods. The Program is open to new Pod Therapy patients coming from multiple daily injections or tubed pumps only who have not previously used Omnipod 5, Omnipod DASH® or Omnipod Insulin Management System.
This offer is not valid for participants whose Omnipod 5 or compatible CGM prescription is paid for in whole or in part by Medicare, Medicaid, or any other federal or state programs. It is not valid for cash-paying participants or where prohibited by law. A participant is considered cash-paying where the participant has no insurance coverage for Omnipod 5 or where the participant has commercial or private insurance but Insulet determines in its sole discretion the participant is effectively uninsured because such coverage does not provide a material level of financial assistance for the cost of an Omnipod 5 prescription. Participants on certain commercial insurance plans may not be eligible. This offer is only valid in the United States, Puerto Rico, and the U.S. territories. Participants receiving their products through the Durable Medical Equipment or Pharmacy Durable Medical Equipment channel are not eligible to participate in the copay card program. Please contact Insulet Customer Support at 1-800-591-3455 for details.
2. Program Details
With this program, Participants may be eligible to receive a limited supply of Omnipod 5 products at no cost for them. Eligible participants have two (2) options, based on the following:
A participant shall sign the Omnipod® 5 Intro Kit 30-Day Trial Acknowledgement through the appropriate platform provided by Insulet.
Once Insulet has received the request, the request shall be escalated to Insulet’s pharmacy partner, where a request for a prescription shall be sent to the participant’s healthcare professional. If a valid prescription is received, both for the Omnipod 5 Intro Kit and the Omnipod 5 Pods, the participant’s benefits will be checked by Insulet or its partners.
a) IF the benefits check results in a monthly copay equal to or below two hundred dollars ($200), then Insulet shall issue a one-time only copay card to the participant, for a value equal to the out-of-pocket expenses the participant would have to pay for an Omnipod 5 Intro Kit, in accordance with Section 3, below.
b) IF the benefits check result in a copay greater than two hundred dollars ($200), Insulet, or its authorized partners, shall arrange for the shipment of one (1) Omnipod 5 Intro Kit, in accordance with Section 4, below.
c)For the purpose of clarity, the term “copay” shall encompass any out-of-pocket expense for one (1) month’s supply of Pods, including any deductible, copays and other out-of-pocket expenses that the participant would have to disburse to procure said supply of Pods.
d)Any copay assistance may not apply to a participant’s health plan’s deductible if prohibited by state law or by a health plan.
e) In order to use the Omnipod 5 System in Automated Mode, the User must also procure a compatible CGM. This program does not include supply of a compatible CGM.
Insulet reserves the right to change, amend or rescind this Program, in whole or in part, at any time.
3. Copay Card
Should participant be deemed eligible to receive an Omnipod 5 Copay Card, participant shall receive electronically one (1) Omnipod 5 Copay Card, valid for a single use, in the amount required for the participant to procure one (1) Omnipod 5 Intro Kit, which shall include:
- One (1) Omnipod 5 Controller
- Ten (10) Omnipod 5 Pods
- One (1) Omnipod 5 User Guide
- One (1) Controller charging cable
4. Product Dispense
Should participant be deemed eligible to receive a one-time dispense of Omnipod 5 Pods at no cost to them, Insulet, or its authorized partner, shall dispense one (1) Omnipod 5 Intro Kit, which shall include:
- One (1) Omnipod 5 Controller
- Ten (10) Omnipod 5 Pods
- One (1) Omnipod 5 User Guide
- One (1) Controller charging cable
The Omnipod 5 Intro Kit shall be delivered to the shipping address indicated by participant in their Acknowledgment Form. Any estimate date of delivery is given solely for participant’s information and does not constitute a warranty that the Intro Kit will be delivered on said date. Participant is responsible to provide an accurate delivery address, to receive shipment of the Intro Kit and to verify the content of the Intro Kit.
Risk Statement
The Omnipod 5 Automated Insulin Delivery System is indicated for use by individuals with type 1 diabetes mellitus in persons 2 years of age and older and type 2 diabetes mellitus in persons 18 years of age and older.
The Omnipod 5 System is intended for single patient, home use and requires a prescription. The Omnipod 5 System is compatible with the following U-100 insulins: NovoLog®, Humalog®, Admelog®, and Kirsty®.
The Omnipod 5 ACE Pump (Pod) is intended for the subcutaneous delivery of insulin, at set and variable rates, for the management of diabetes mellitus in persons requiring insulin. The Omnipod 5 ACE Pump is able to reliably and securely communicate with compatible, digitally connected devices, including automated insulin dosing software, to receive, execute, and confirm commands from these devices.
SmartAdjust™ technology is intended for use with compatible integrated continuous glucose monitors (iCGM) and alternate controller enabled (ACE) pumps to automatically increase, decrease, and pause delivery of insulin based on current and predicted glucose values.
The Omnipod 5 SmartBolus Calculator is intended to calculate a suggested bolus dose based on user-entered carbohydrates, most recent sensor glucose value (or blood glucose reading if using fingerstick), rate of change of the sensor glucose (if applicable), insulin on board (IOB), and programmable correction factor, insulin to carbohydrate ratio, and target glucose value.
WARNING: SmartAdjust technology should NOT be used by anyone under the age of 2 years old. SmartAdjust technology should also NOT be used in people who require less than 5 units of insulin per day as the safety of the technology has not been evaluated in this population.
The Omnipod 5 System is NOT recommended for people who are unable to monitor glucose as recommended by their healthcare provider, are unable to maintain contact with their healthcare provider, are unable to use the Omnipod 5 System according to instructions, are taking hydroxyurea as it could lead to falsely elevated CGM values and result in over-delivery of insulin that can lead to severe hypoglycemia, and do NOT have adequate hearing and/or vision to allow recognition of all functions of the Omnipod 5 System, including alerts, alarms, and reminders. Device components including the Pod, CGM transmitter, and CGM sensor must be removed before Magnetic Resonance Imaging (MRI), Computed Tomography (CT) scan, or diathermy treatment. In addition, the Controller and smartphone should be placed outside of the procedure room. Exposure to MRI, CT, or diathermy treatment can damage the components. Visit www.omnipod.com/safety for additional important safety information.
WARNING: DO NOT start to use the Omnipod 5 System or change settings without adequate training and guidance from a healthcare provider. Initiating and adjusting settings incorrectly can result in over-delivery or under-delivery of insulin, which could lead to hypoglycemia or hyperglycemia.