URGENT: Medical Device Correction UPDATE
REF: 9056196-03/12/2026-001-C
Please read this communication in its entirety and click below.
Urgent Information Regarding the Omnipod® 5 Pod
Affected Product
| Products Impacted: | Omnipod® 5 Pods |
| Lot Numbers: | See list in Appendix B for a list of affected Lot numbers or check if your Pods are affected |
| UDI: | 10385083000527 |
Insulet is providing an update to the voluntary Medical Device Correction that was initiated on March 12, 2026 for specific lots of Omnipod® 5 Pods due to a manufacturing issue we identified through our ongoing product monitoring. Based on our investigation, this issue is isolated to identified lots distributed in the United States only.
What is the new information being provided?
Out of abundance of caution, we are providing the list of affected lots, expanding the list to include Pods that have expired, and providing additional details below on the Pod alert that you may experience with this issue. Pods not included in these lots, as well as all other Omnipod® 5 Pods and Omnipod® products remain safe to use as intended.
The U.S. Food and Drug Administration (FDA) has been notified of this action.
What is the Issue?
Insulet identified that some Pods from specific lots may have a small tear in the internal tubing that delivers insulin. If this happens, insulin may leak inside the Pod instead of being fully infused in the body as intended potentially leading to underdelivery of insulin.
If there is a fluid leak inside the Pod, you may receive a hazard alarm to notify you to remove your Pod. In some cases, there may be no alarm or alert, and the issue may go unnoticed, potentially leading to underdeliver of insulin with the potential for prolonged high blood glucose levels. If this occurs, your blood glucose levels may increase and may not respond as expected to additional insulin delivery, which may result in an Automated Delivery Restriction alert advising you to troubleshoot.
Because this problem may happen without triggering an alarm or alert, you could receive less insulin than needed without realizing it. For this reason, do not rely only on alarms to know if something is wrong. If you have Pods from an affected lot, switch to a Pod from a nonaffected lot as soon as possible or talk with your healthcare provider about other options for insulin delivery.
This issue does not affect continuous glucose monitoring (CGM) systems or CGM readings.
Potential Risks:
If insulin is not delivered properly, users may experience high blood glucose levels due to underdelivery of insulin. In the most severe cases, prolonged and persistent high blood glucose levels can lead to diabetic ketoacidosis (DKA), a serious medical condition that requires prompt medical treatment and can be life-threatening if not treated. The risk of underdelivery increases if you use more than one affected Pod in a row.
Insulet has received 29 reports of serious adverse events associated with high blood glucose levels, including hospitalization and diabetic ketoacidosis. No deaths have been reported.
Please take the following steps immediately:
- DO NOT use Pods from the affected lots. Affected lots are listed below in Appendix B.
- It is important that you check the lot number on your Pod tray lid, the Pod box or the Pod itself to determine if your Pods are affected.
- Provided in Appendix A are instructions and pictures to help you identify the lot number for your pods OR
- Visit this page to check if your Pods are affected.
All Pods within a single box will be from a single lot and if the lot is affected, do not use any of those Pods.
- DO NOT use Pods from the affected lots.
- Discontinue use of any impacted Pod immediately. If the Pod you are currently using is from an affected lot, it is important that you immediately change your Pod to resume insulin delivery. When changing your Pod, confirm that the new Pod is not from an affected lot.
- You can request a return/replacement of any unused affected Pods through the following options:
- Log in to your PodderCentral account to complete the self-serve return/replacement form.
Connect with a live agent via chat by clicking the Podder Support button in the lower right of this page or visit https://www.omnipod.com/current-podders
Call our dedicated Product Support phone line at 1-800-641-2049, available 24/7.
Always check the expiration date before using a Pod. Do not use any Pod that is past its expiration date, as expired Pods may not function as intended.
If Pod supply is depleted due to this issue, consult with your healthcare provider for alternative means of insulin delivery while waiting for Pods to be replaced. Always follow your healthcare provider's guidance on appropriate glucose monitoring.
What is Insulet doing to address this issue?
Following our investigation, we have implemented updates to our manufacturing processes and quality controls to strengthen detection and prevention of this issue and further support the integrity of our products.
We also have sufficient supply available to replace affected Pods and do not anticipate any disruption to product availability.
Additional information:
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail or by fax. Complete and submit the report online at www.fda.gov/medwatch. Regular Mail or Fax: Download form or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800-FDA-0178.
For additional support, please connect with a live agent by selecting the Podder Support button on the bottom right corner or call our dedicated Product Support phone line at 800-641-2049, available 24/7.
We understand how much you depend on your Omnipod every day. We sincerely regret the concern and disruption this may cause and are committed to supporting you. Thank you for taking the time to review this important information and for the trust you place in Omnipod.
Sincerely,

Chuck Medovich
Senior Vice President, Quality Assurance, Regulatory Affairs, and Compliance
Insulet Corporation
Appendix A: How to locate lot number:
The lot number can be located on the Pod tray lid, the flat side of the Pod, as well as the 5-pack Pod box.
Pod lot number location on the Pod tray lid (circled below in red):
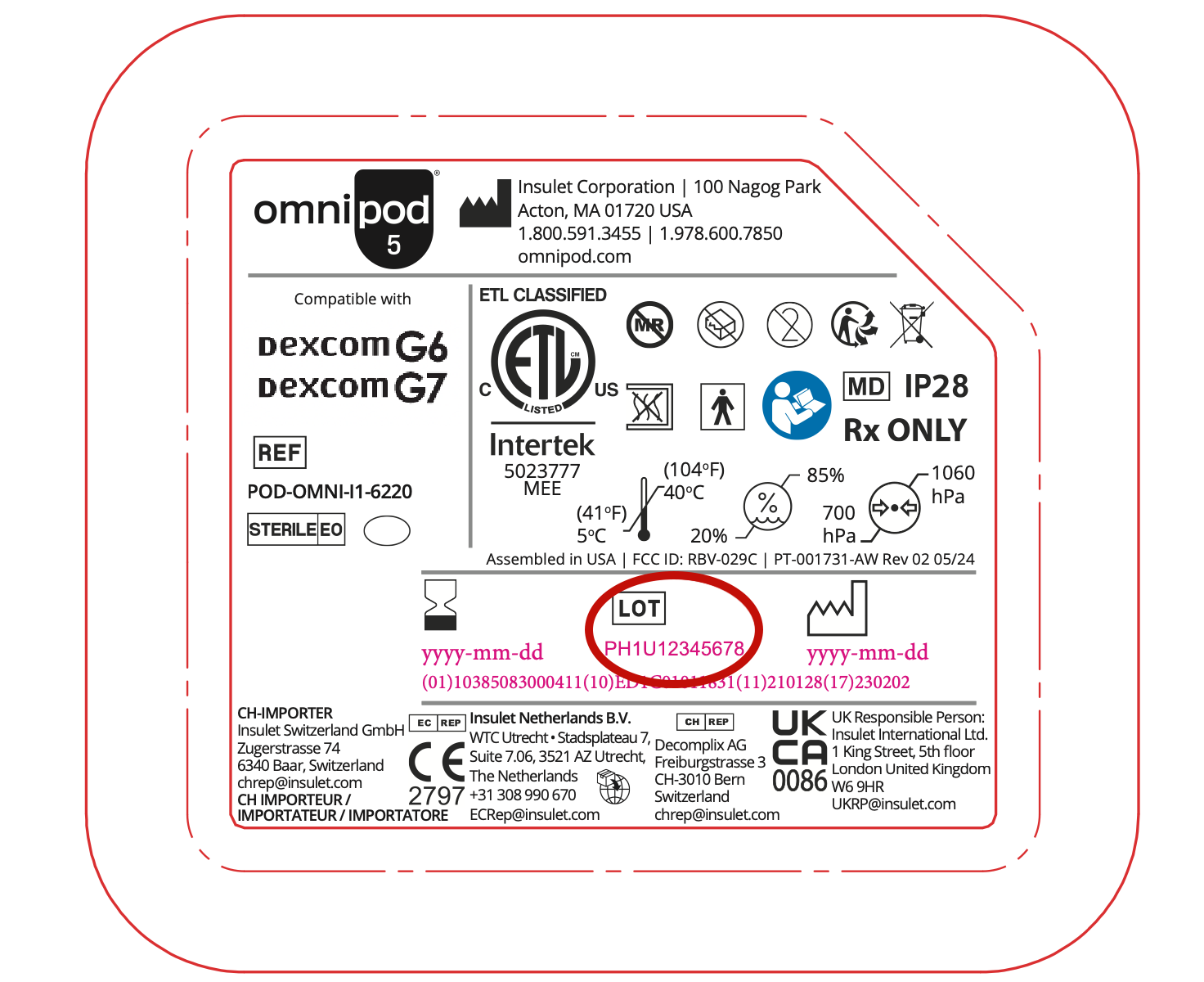
Pod lot number location on the side of the 5-pack Pod box: 

Pod lot number location on the Pod itself (flat side):

Appendix B: Affected Pod Lots
The list of Omnipod 5 Pod lot numbers affected by this Medical Device Correction are listed in the table below, along with their expiration date. Lots highlighted in Yellow are past expiration.
| Pod Lot Number | Expiration Date |
| PH1U10242521 | 10/24/27 |
| PH1U10242531 | 10/24/27 |
| PH1U10242541 | 10/24/27 |
| PH1U10232531 | 10/23/27 |
| PH1U10232541 | 10/23/27 |
| PH1U10222531 | 10/22/27 |
| PH1U10222541 | 10/22/27 |
| PH1U10212531 | 10/21/27 |
| PH1U10212541 | 10/21/27 |
| PH1U10202511 | 10/20/27 |
| PH1U10202521 | 10/20/27 |
| PH1U10202531 | 10/20/27 |
| PH1U10202541 | 10/20/27 |
| PH1U10182531 | 10/18/27 |
| PH1U10182541 | 10/18/27 |
| PH1U10172531 | 10/17/27 |
| PH1U10172541 | 10/17/27 |
| PH1U10162531 | 10/16/27 |
| PH1U10162541 | 10/16/27 |
| PH1U10152541 | 10/15/27 |
| PH1U09252521 | 09/25/27 |
| PH1U09252531 | 09/25/27 |
| PH1U09242511 | 09/24/27 |
| PH1U09242521 | 09/24/27 |
| PH1U09242531 | 09/24/27 |
| PH1U08182531 | 08/18/27 |
| PH1U08182541 | 08/18/27 |
| PH1U08162531 | 08/16/27 |
| PH1U05052511 | 11/05/26 |
| PH1U03312511 | 10/01/26 |
| PH1U03312521 | 10/01/26 |
| PH1U04012511 | 10/01/26 |
| PH1U04012521 | 10/01/26 |
| PH1U03282511 | 09/28/26 |
| PH1U03282522 | 09/28/26 |
| PH1U02252541 | 08/25/26 |
| PH1U08062421 | 02/06/26 |
| PH1U08062411 | 02/06/26 |
| PH1U08052431 | 02/05/26 |
| PH1U08032411 | 02/03/26 |
| PH1U08032421 | 02/03/26 |
| PH1U07252421 | 01/25/26 |
| PH1U05162431 | 11/16/25 |
| PH1U05162421 | 11/16/25 |
| PH1U05162411 | 11/16/25 |
| PH1U05152421 | 11/15/25 |
| PH1U05152411 | 11/15/25 |
| PH1U05152431 | 11/15/25 |
| PH1U03212421 | 09/21/25 |
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA’s MedWatch Adverse Event Reporting program either online, by regular mail or by fax. Complete and submit the report online a www.fda.gov/medwatch. Regular Mail or Fax: Download form or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form or submit by fax to 1-800-FDA-0178.
We understand how much you depend on your Omnipod every day. We sincerely regret the concern and disruption this may cause and are committed to supporting you.
For additional support, please connect with a live agent by selecting the “Podder Support” button on the bottom-right corner or call Product Support at 800-641-2049, available 24/7.
Frequently Asked Questions
I saw the FDA recently posted something on their website. Is this a new issue?
Why did Insulet issue a Medical Device Correction for Omnipod 5 Pods?
What could happen if a Pod from an affected lot is used?
Has the FDA been notified?
How do I know if my Omnipod 5 Pods are affected?
What if I already used an affected Omnipod 5 Pod?
Who can I call if I have any questions?
How is Insulet communicating this issue to customers?
Has anyone been hurt due to this issue with Omnipod 5?
Which Omnipod products are affected by this voluntary Medical Device Correction?
Does this affect Omnipod users in international markets?
Will Insulet continue to ship Omnipod 5 Pods?
How long has Insulet known there was an issue with Omnipod 5? How was the issue discovered?
Is the issue with the Omnipod 5 App/Controller or with the Omnipod 5 Pods?
What steps is the company taking to correct the problem and ensure it doesn’t happen again?
I received the notice of voluntary Medical Device Correction from Insulet. Do I have to do anything?
I believe I may have Omnipod 5 Pods that are impacted by this issue. How do I get replacements?
How do I find the Lot number?
One of my Omnipod 5 Pods is part of the affected lot. Is it safe to use another Omnipod 5 Pod from the same box?
What if none of my Omnipod 5 Pods are included in the affected lots? Is it safe to continue using unaffected Omnipod 5 Pods?
If I get an alarm on my Omnipod 5 Pod, does this mean I have a faulty Pod and need a replacement?
If I have Omnipod 5 Pods that are affected, will Insulet exchange them for Omnipod 5 Pods that are not affected?
How long will it take to receive a replacement Omnipod 5 Pod?
What do I do if all my Pods are affected?
Do I need to return affected Omnipod 5 Pods? How do I do that?
Is Insulet paying for the return of my Omnipod 5 Pods?
Will I need a new prescription (Rx) or additional copay to obtain replacement Omnipod 5 Pods?
My Omnipod 5 Pods are affected, and I need to get new Omnipod 5 Pods. Will my insurance allow an early refill and will I have to pay my copay to get replacement Pods?