Omnipod® 5

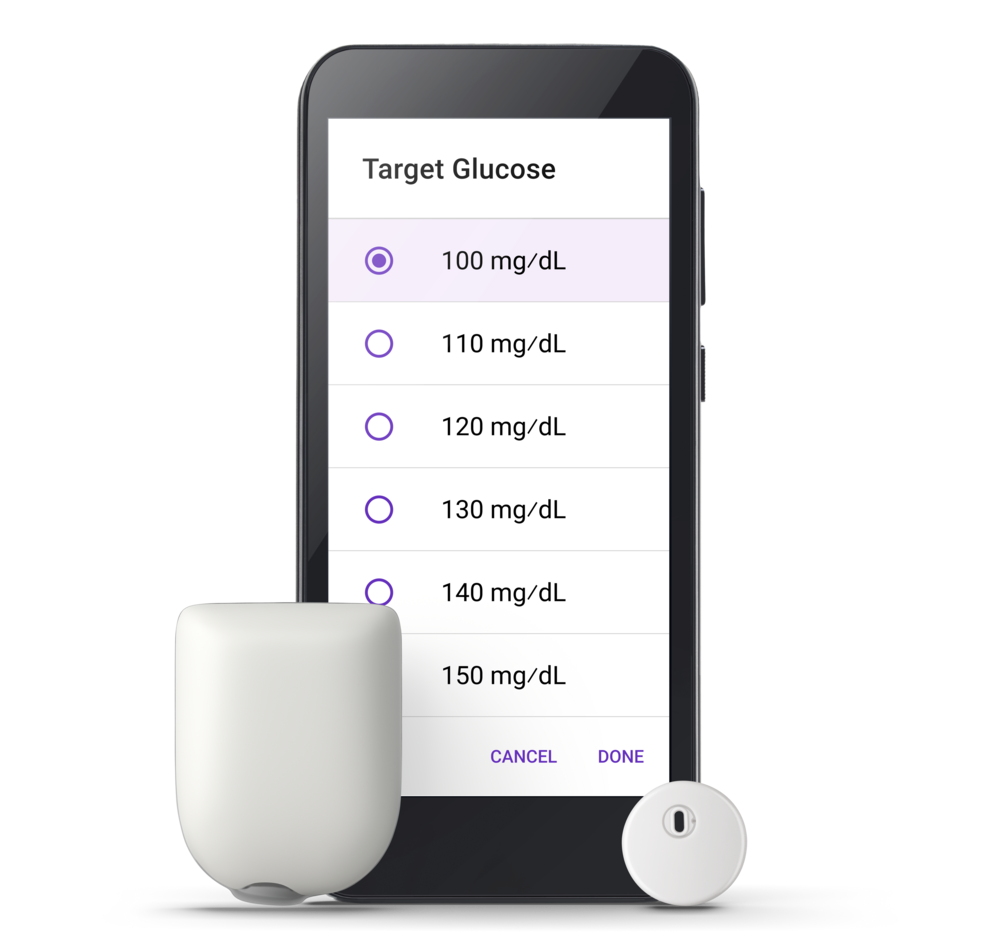

A powerful update is coming soon
Omnipod 5 is about to get stronger with an enhanced algorithm and expanded sensor capability.
- New 100 mg/dL Target Glucose for more time in range, with no meaningful impact on time below range1
- Updated alarm handling designed for fewer interruptions and more time in Automated Mode
- Omnipod 5 will soon integrate with the FreeStyle Libre 3 Plus sensor - providing more sensor choice and individualized care for your patients
Transform T1 and T2 Diabetes Care with Omnipod 5 AID
Omnipod 5 is the first and only tubeless automated insulin delivery system in the US market. Give your patients freedom from tubes with a modern, wearable insulin management Pod with demonstrated strong glycemic results.2-4
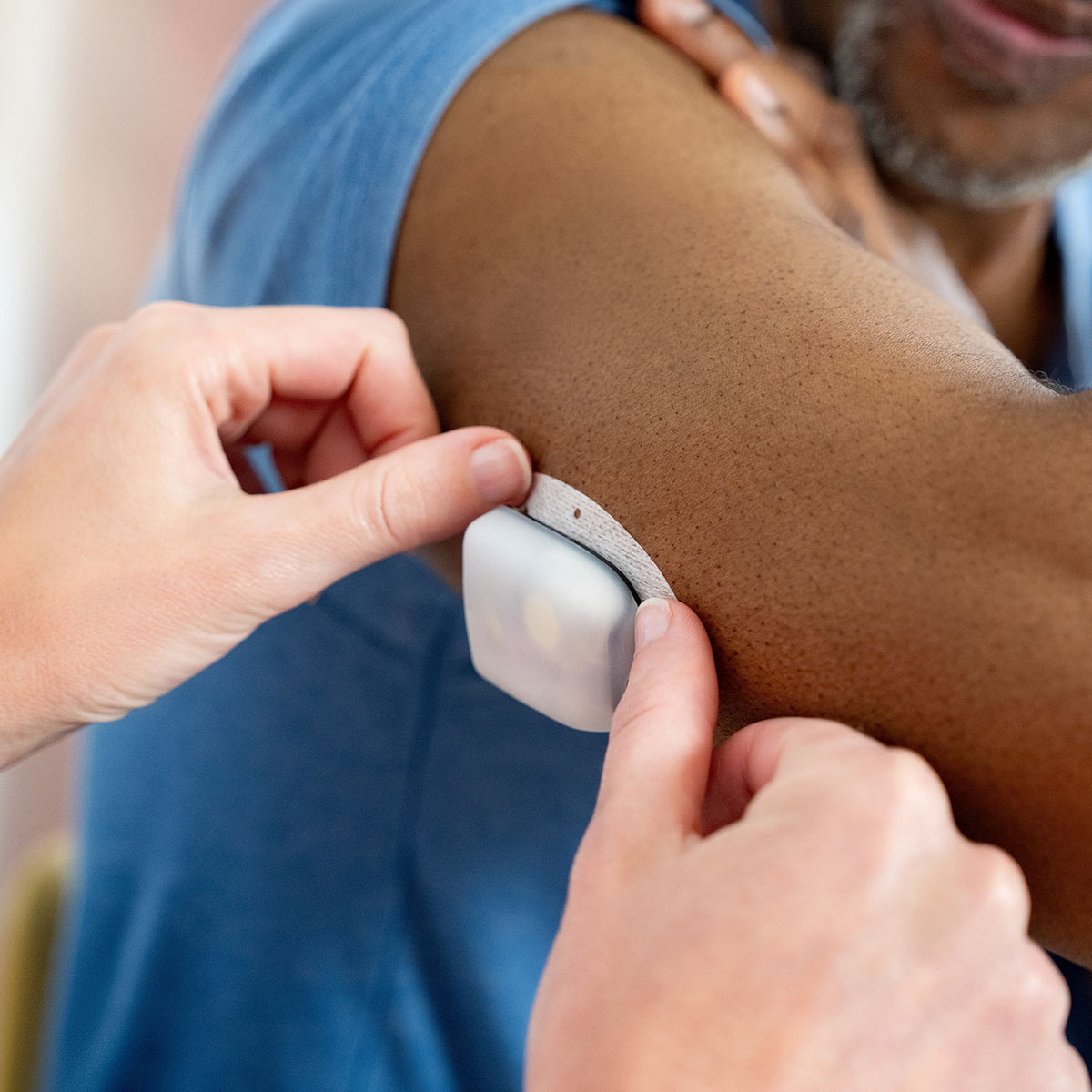

Who It’s For:
A Powerful Pod Designed for Every Diabetes Journey
- Indicated for type 1 diabetes for ages 2+, and type 2 diabetes for ages 18+
- Ideal for adults and pediatrics to switch from multiple daily injections (MDI), aligned to ADA standards for AID as the preferred insulin delivery method over MDI for type 1 and type 2 diabetes5
- Discreet, waterproof6, on-body Pod with less interactions and less hassles7
- Compatible with leading CGM sensors including Dexcom G7 Series*, FreeStyle Libre 2 Plus sensors, and soon to be FreeStyle Libre 3 Plus sensor, so more patients have the freedom to choose their sensor
- Over 90% insured patients nationwide are covered for the Omnipod 58
- Find out now if your patients are covered
Outstanding Clinical Results: Setting the Standard for Real-World AID
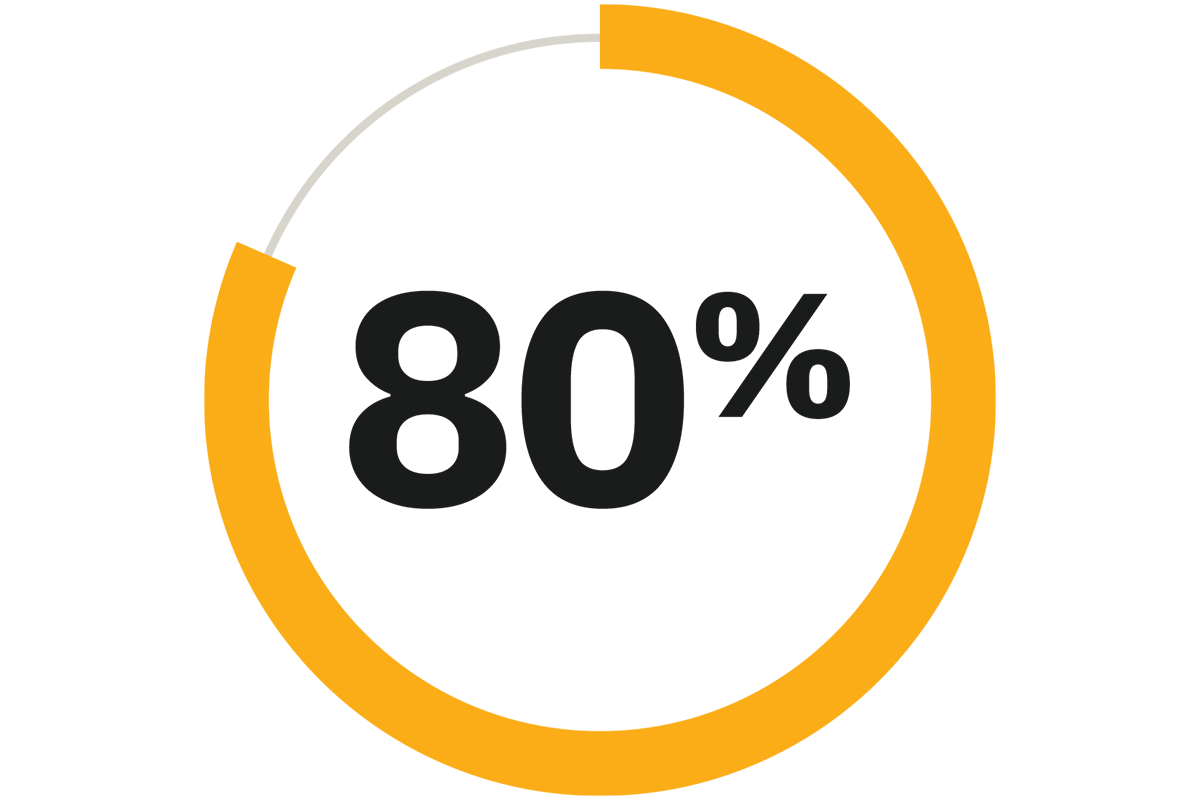

Time in range (TIR) with optimized settings9
Maximize TIR for your patients with a simplified diabetes management system. Omnipod 5 real-world evidence demonstrated that type 1 and type 2 patients achieved over 80% time in range (TIR) with optimized settings.9
Omnipod 5 SECURE-T2D Pivotal Trial
Omnipod 5 RADIANT Randomized Controlled Trial
What Users Are Saying About Omnipod 512
- 96% of users agree, Omnipod 5 is easy to use
- 92% of users agree, they trust Omnipod 5
- 91% of users agree, they would not go back to tubes
- 96% of users agree, they love the automatic cannula insertion feature
Hear from colleagues who not only prescribe Omnipod 5, but use it themselves.
How Does the Omnipod 5 Algorithm Work?
Omnipod 5 automatically adjusts insulin delivery every 5 minutes to help keep users in range (and help to correct highs and protect from lows,) day and night.2-4
When the system predicts rises in glucose levels, it can deliver corrections up to 4x of the patient’s automated rate, for up to 12 corrections per hour. When glucose is trending down, Omnipod 5 can also proactively decrease or pause insulin delivery to help protect against hypoglycemia.2-4
With each Pod change, Omnipod 5 further personalizes care by calculating a new adaptive rate based on the patient’s evolving total daily insulin (TDI) requirements.1
Want to get a hands-on understanding of our intuitive Omnipod 5 AID system?
Try the free simulator app on your smartphone.
Ready to Prescribe?
We offer not only a streamlined prescribing process through the pharmacy, but provider resources to help you support your patients.
*The Dexcom G7 15 Day CGM is indicated for adults (18+)
Dexcom is in the process of discontinuing Dexcom G6 to focus on delivering the next generation of CGM. Omnipod 5 will continue to support integration with Dexcom G6 for as long as this sensor remains available. For more information, please visit www.dexcom.com or contact Dexcom support.
The Omnipod 5 Automated Insulin Delivery System is indicated for use by individuals with type 1 diabetes mellitus in persons 2 years of age and older and type 2 diabetes mellitus in persons 18 years of age and older. The Omnipod® 5 System is intended for single patient, home use and requires a prescription. The Omnipod® 5 System is compatible with the following U-100 insulins: NovoLog®, Humalog®, Kirsty®, and Admelog®. Warning: SmartAdjust™ technology should NOT be used by anyone under the age of 2 years old. SmartAdjust™ technology should also NOT be used in people who require less than 5 units of insulin per day as the safety of the technology has not been evaluated in this population. Refer to the Omnipod® 5 Automated Insulin Delivery System User Guide and www.omnipod.com/safety for complete safety information including indications, contraindications, warnings, cautions, and instructions. The sensor housing, FreeStyle, Libre, and related brand marks are marks of Abbott and used with permission. For full FreeStyle Libre 3 Plus Sensor safety information, please visit www.freestyle.abbott/us-en/safety-information
1. In a simulated analysis, the 100 mg/dL Target Glucose (TG) setting demonstrated superior Time in Range (70-180 mg/dL) and non-inferior Time Below Range (<70 mg/dL) compared with results at the 110-150 mg/dL TG settings in a real-world population of people with T1D using Omnipod 5. Differences in mean TIR for 100mg/dL vs 110mg/dL, vs 120 mg/dL, vs 130 mg/dL, vs 140 mg/dL, vs 150 mg/dL were 2.5%, 4.8%, 9.8%, 15.3%, and 20.8%, respectively. For each comparison, the lower bound of the one-sided 95% Confidence Interval (CI) was > 0, meeting the pre-specified superiority criterion. Differences in mean TBR for 100mg/dL vs 110mg/dL, vs 120 mg/dL, vs 130 mg/dL, vs 140 mg/dL, vs 150 mg/dL were 0.4%, 0.7%, 0.8%, 0.9%, and 0.9%, respectively. For each comparison, the upper bound of the one-sided 95% Confidence Interval (CI) was below the 2% non-inferiority margin. Analysis not controlled for ISF, ICR, % time in Automated Mode and number of bolus/day. Data on File. RF-012026-00057.
2. Brown S. et al. Diabetes Care. 2021;44:1630-1640. Prospective pivotal trial in 240 participants with T1D aged 6 - 70 yrs [adults/adolescents (n= 128; aged 14-70 yrs) children (n=112; aged 6-13.9 yrs)]. Study included a 14-day standard therapy (ST) phase followed by a 3-month Omnipod 5 hybrid closed-loop phase. Mean time >180 mg/dL in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 32.4% vs. 24.7%; 45.3% vs. 30.2%, P<0.0001, respectively. Mean time <70 mg/dL in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 2.9% vs. 1.3%, P<0.0001; 2.2% vs. 1.8%, P=0.8153, respectively. Mean time 70-180 mg/dL as measured by CGM in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 64.7% vs. 73.9%; 52.5% vs. 68.0%, P<0.0001, respectively. Results measured by CGM. Mean HbA1c in adults/adolescents and children, ST vs. 3-mo Omnipod 5: 7.16% vs 6.78%, P<0.0001; 7.67% vs 6.99%, P<0.0001, respectively.
3. Sherr JL, et al. Diabetes Care. 2022. 45(8):1907–1910. Prospective trial in 80 participants with T1D aged 2 - 5.9 yrs. Study included a 14-day standard therapy (ST) phase followed by a 3-month Omnipod 5 hybrid closed-loop (HCL) phase. Mean time >180 mg/dL in very young children (2 - 5.9yrs) as measured by CGM: ST = 39.4%, 3-mo Omnipod 5 = 29.5%, P<0.0001. Mean time <70 mg/dL in very young children (2-5.9 yrs) as measured by CGM: ST = 3.43%, 3-mo Omnipod 5 = 2.46%, P=0.0204. Results measured by CGM. Mean time in range (70-180mg/dL) in very young children (2 - 5.9 yrs) as measured by CGM: ST = 57.2%, 3-mo Omnipod 5 = 68.1%, P<0.05. Mean HbA1c: ST vs. Omnipod 5 use in very young children (2 - 5.9 yrs) 7.4% vs 6.9%, P<0.05.
4. Pasquel FJ, et al. JAMA Network Open (2025). Prospective pivotal trial in 305 participants with T2D aged 18-75 yrs. Study included a 14-day standard therapy (ST) phase followed by a 13-week Omnipod 5 hybrid closed-loop phase. Mean time >180 mg/dL as measured by CGM: ST = 54%, 3-mo Omnipod 5 = 34%, P<0.001. Mean time <70 mg/dL as measured by CGM: ST = 0.2%, 3-mo Omnipod 5 = 0.2%. Mean time in range (70-180 mg/dL): ST vs. 13-week Omnipod 5: 45% vs. 66%, P<0.001. Mean HbA1c: ST vs. 13-week Omnipod 5: 8.2% vs. 7.4%, P<0.001. 53 participants used carbohydrate counting pre-study with mean HbA1c: ST vs. 13-week Omnipod 5: 7.8% vs. 7.3%. 129 participants used fixed dose pre study with mean HbA1c: ST vs. 13-week Omnipod 5: 8.4% vs. 7.4%. 46 participants used sliding scale pre study with mean HbA1c: ST vs. 13-week Omnipod 5: 8.1% vs. 7.3%. 32 participants used set doses to bolus pre study with mean HbA1c: ST vs. 13-week Omnipod 5: 8.3% vs. 7.4%, P=0.85. 36 participants used other carb counting method pre study with mean HbA1c: ST vs. 13-week Omnipod 5: 8.5% vs. 7.4%, P=0.85. Mean total daily dose (TDD): ST = 0.8U/kg/day, 3-mo Omnipod 5 = 0.57U/kg/day, P<0.001. Comparison is relative change. Mean T2-DDAS total intensity score: ST = 2.5, 3-mo Omnipod 5 = 2.2, P<0.001. Mean Proportion with T2-DDAS ≥ 2.0, no. (%): ST = 66%; 3-mo Omnipod 5 = 55%, P<0.001.
5. American Diabetes Association Professional Practice Committee for Diabetes. 7. Diabetes technology: Standards of Care in Diabetes—2026. Diabetes Care 2026;49 (Suppl. 1):
6. The Pod has an IP28 rating for up to 7.6 m or 25 feet for 60 minutes. The PDM is not waterproof.
7. The Omnipod 5 Pod is tubeless and waterproof, designed to hold up to 200 units of insulin and replaces an average of 14 injections delivering insulin for up to 72 hours.
8. Reflects coverage for Omnipod 5 DexG7G6 Pods in commercial channel only. Source: Managed Markets Insights & Technology, LLC. Data as of January 2026
9. Retrospective RWE data on file. 2025. Results shown for users with optimized settings including sufficient CGM data (≥75% of days with ≥220 readings), ≥90% time in Automated Mode, ≥5 bolus/day and an average Target Glucose of 110-115 mg/dL. Optimized settings: ISF x TDI ≤1500, I:C Ratio x TDI ≤350. RF-062025-00014.
10. Pasquel FJ, et al. JAMA Network Open (2025). Race/ethnicity of participants reported as 24% Black, 22% Latino/Hispanic. Comparison based on published clinical trials of the current AID landscape in T2D. Insulet Data on File. RF-082024-00024
11. Wilmot E, et al. Lancet (2026); A 13-week randomized, parallel-group clinical trial conducted among 188 participants (age 4-70) [51% HbA1c≥ 8%] with type 1 diabetes in France, Belgium, and the U.K., comparing the safety and effectiveness of the Omnipod 5 System versus multiple daily injections with CGM. Time in range (70-180 mg/dL) with OP5 improved by 22% (43% MDI vs. 65% OP5, p<0.001). Time below range (<70 mg/dL) with OP5 was non-inferior to MDI (2.27% MDI vs. 2.56%, p=0.2). Mean time in range (70-180 mg/dL) improvement of 26% on day 1 of using Omnipod 5. Data on file. RF-072025-00023
12. 2025 Customer Survey of U.S. adults with Type 1 and Type 2 diabetes using Omnipod 5, (n=1,532). 96% agreed Omnipod 5 is easy to use. 92% agreed they trust Omnipod 5. 91% agreed would not go back to tubes. 96% agreed they love the automatic cannula insertion feature. Data on file. RF-082025-00031.
13. In Automated Mode, SmartAdjust technology uses total daily insulin (TDI) to set a new Adaptive Rate. Omnipod 5 User Manual. P. 291
INS-OHS-02-2023-00071 V 10.0