Hybrid Closed Loop (HCL), explained.
Hybrid Closed Loop (HCL), also known as Automated Insulin Delivery (AID), is revolutionising the way people manage their type 1 diabetes.
What is Hybrid Closed Loop System?
AID systems, also known as Hybrid Closed Loop (HCL) systems, use an insulin pump and a compatible glucose sensor that ‘communicate’ to automatically deliver insulin based on real-time glucose information.
AID uses smart software known as a control algorithm to frequently adjust basal insulin doses in small amounts, continuously calculating how much insulin to deliver as the user’s glucose levels fluctuate.
With this precise and personalised dosing, automated insulin delivery can improve glucose control and reduce A1c, in addition to reducing some of the burden of diabetes management1-3.


How does Hybrid Closed Loop system work?
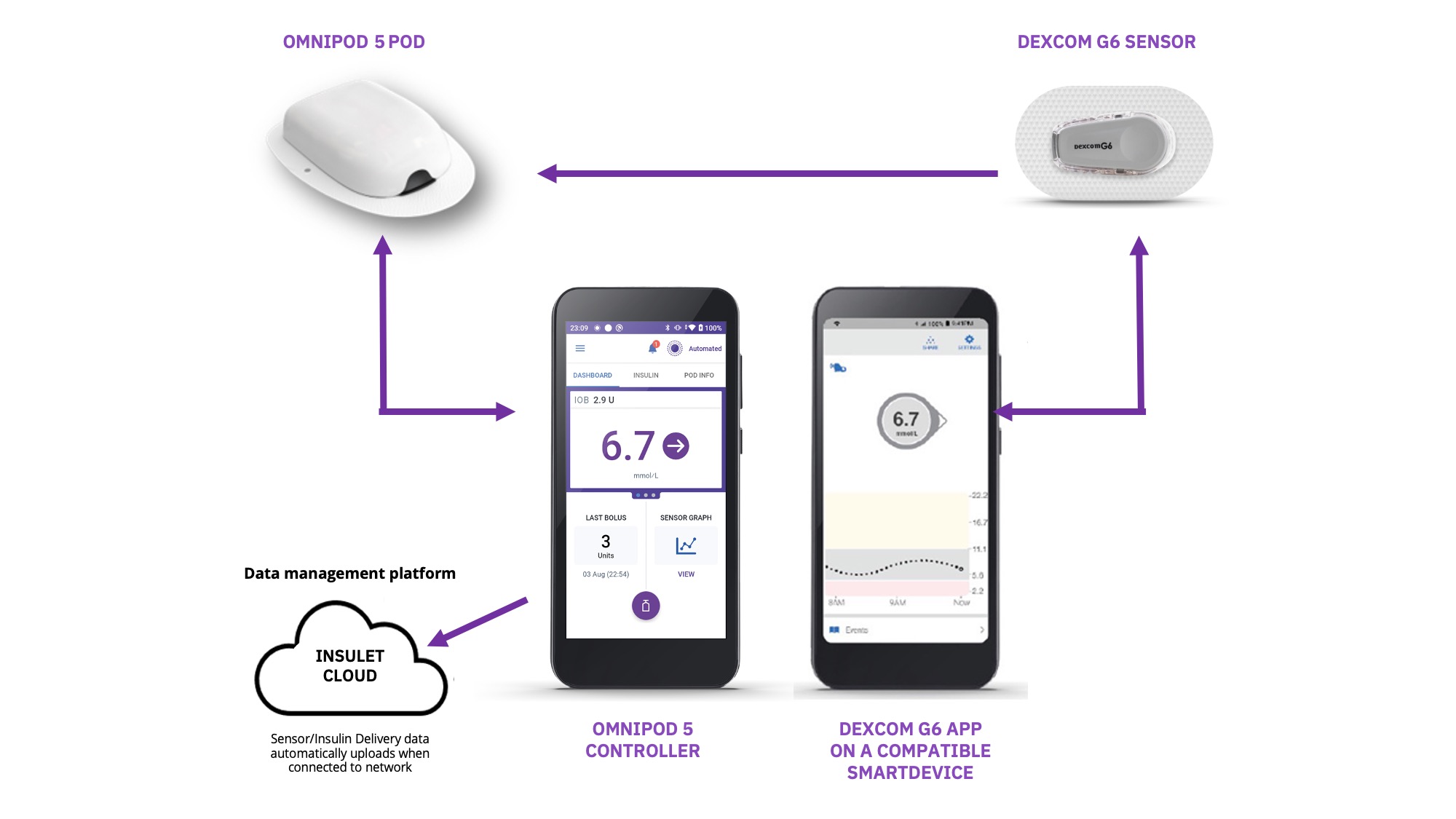
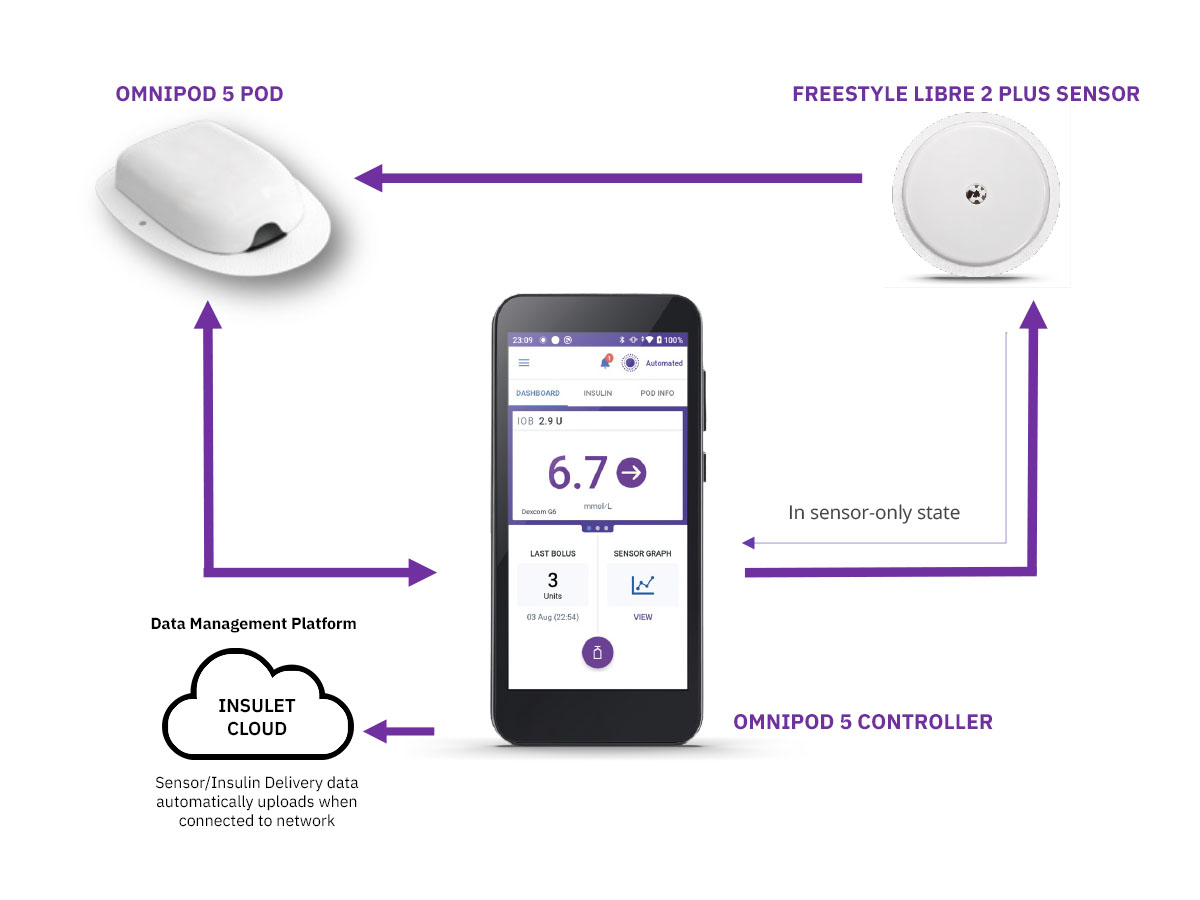
An automated insulin delivery (AID) or Hybrid Closed Loop (HCL) system is made up of three main parts:
- An insulin pump or Pod
- A compatible glucose sensor
- Smart software, known as a control algorithm
Click on below to find out more about insulin pumps and glucose sensors or read on to learn how they work together with the algorithm in an AID system!
AID allows for a complete circle of communication between the Pod or Pump, the glucose sensor and the control algorithm.
In simple terms, the sensor talks to the Pod or Pump, where the algorithm then responds by automatically increasing, decreasing or pausing insulin delivery depending on whether glucose levels are rising or falling.
Over time, the system learns more about your body’s total daily insulin needs to make predictions about your glucose levels. But Hybrid Closed Loop (HCL) AID systems are not fully automatic - you still need to bolus insulin for food.
Hybrid Closed Loop (HCL) in practice
Generally, every 5 minutes:
- The glucose sensor reports your glucose levels to the algorithm.
- The algorithm calculates whether insulin delivery needs to be increased, decreased, remain the same or stopped completely.
- The Pump or Pod actions the calculation.
- The sensor picks up on any changes to your glucose level and reports that to the algorithm… and the circle continues. Which is the reason for the term ‘loop’.

How does this help people with type 1 diabetes?
AID systems are an exciting development in diabetes technology because the automated adjustments to insulin delivery can help to control blood glucose levels, reduce hypoglycaemia (low blood sugars) and increase time in range1-3.
An AID system can help lessen the burden of managing your diabetes, as well as helping to reduce highs and lows throughout the day and night1-3.
There are several automated insulin delivery systems now available, so speak to your diabetes team about your options.
https://www.diabetes.org.uk/guide-to-diabetes/diabetes-technology/closed-loop-systems
https://jdrf.org.uk/information-support/treatments-technologies/continuous-glucose-monitors/hybrid-closed-loop-technology/
https://abcd.care/dtn/educational-resources-people-living-diabetes
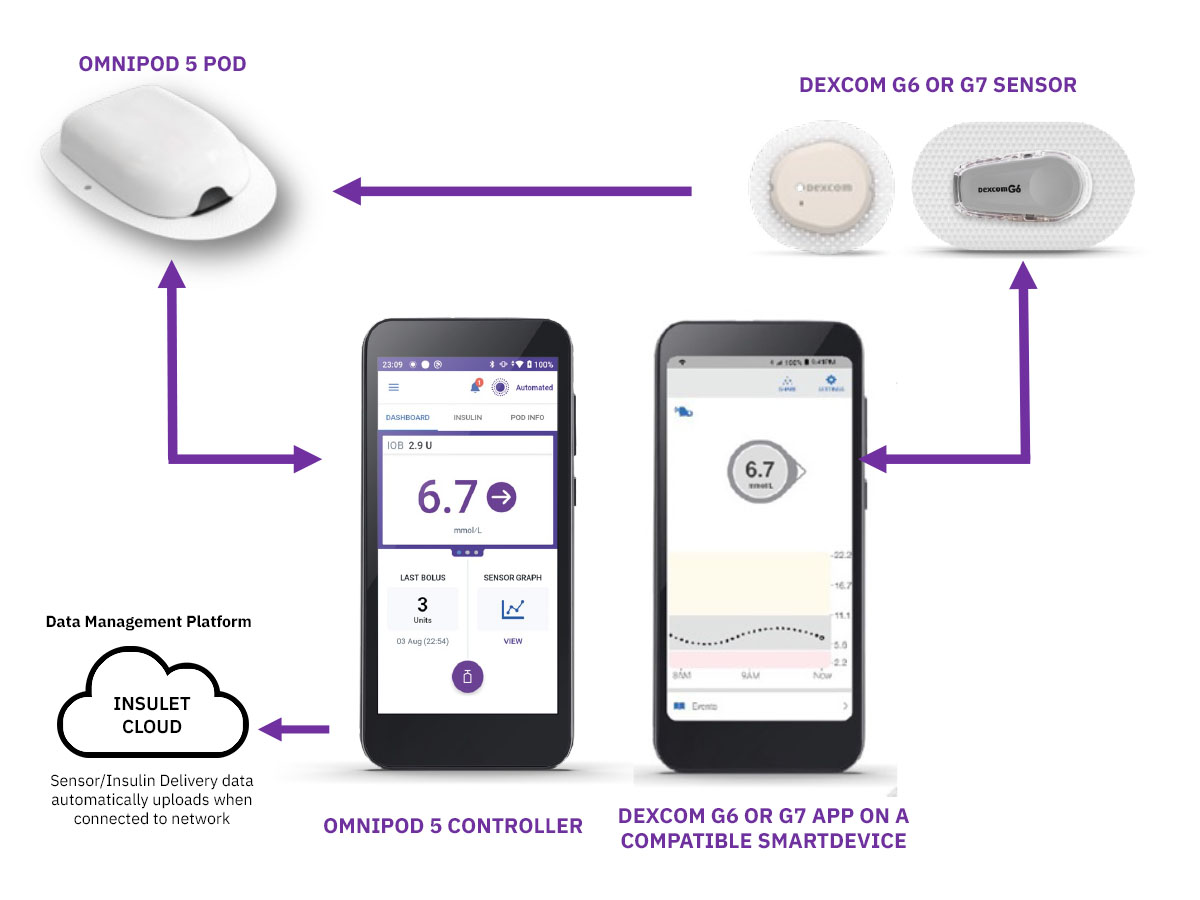
Below, you can learn more about Insulet’s AID System: The Omnipod® 5 Automated Insulin Delivery System.
Say hello to tubeless automated insulin delivery with Omnipod® 5
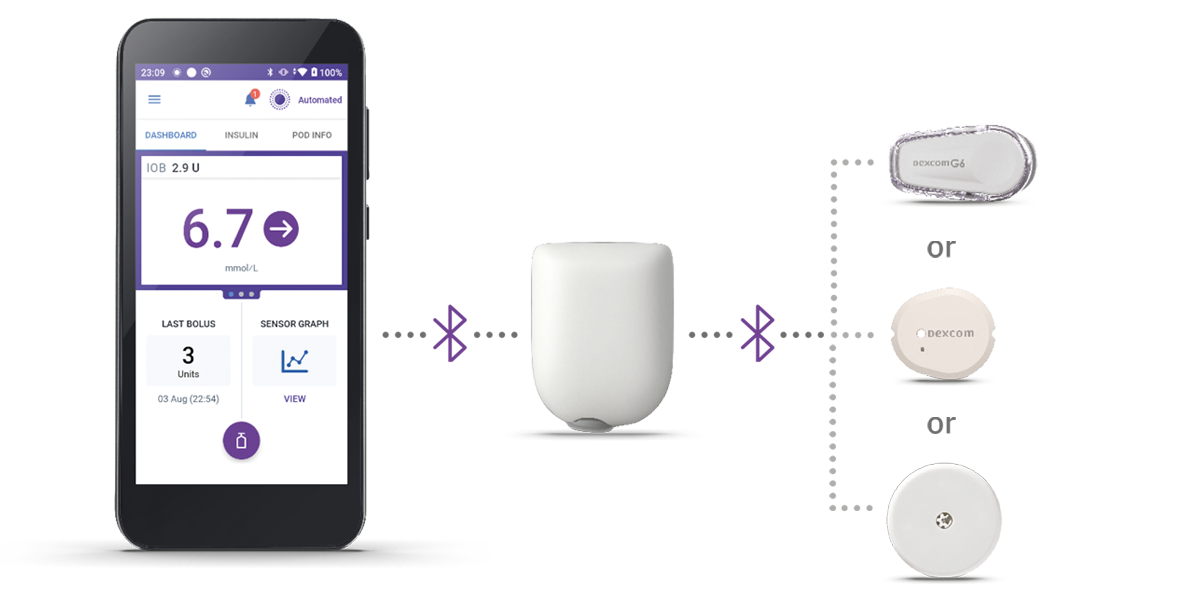
Omnipod 5 is the first and only tubeless AID system integrated with the leading sensor* brands, offering automated† insulin delivery with freedom from multiple daily injections, insulin pump tubing AND finger prick testing§.
Omnipod 5 is approved for people living with type 1 diabetes aged 2 and older.
Automated Insulin Delivery, Simplified.
You choose your target glucose setting. There are five options between 6.1mmol/L and 8.3mmol/L.
Each Pod contains SmartAdjust™ Technology that adjusts insulin every 5 minutes to bring your glucose value to your customized glucose target or target glucose.
Each wearable, waterproof‡ Pod continuously delivers insulin for up to three days (72 hours).
Bolus doses and correction doses are given by using the Omnipod 5 Controller.
The result? More time in range2,3 and improved A1c.2,3


Additional Features of the Omnipod 5 AID System
SmartBolus Calculator.
The SmartBolus Calculator can suggest a bolus amount of insulin based on your individual settings, entered values, and sensor glucose value and trend.
Activity feature.
Enabling the Activity Feature will automatically increase the glucose target and reduce insulin delivery to help keep glucose in range when glucose levels typically go low, for example during exercise.
Is the Omnipod 5 right for me?
Frequently Asked Questions about Hybrid Closed Loop System
What is the Omnipod 5 Automated Insulin Delivery System?
Is an AID system the same as a Hybrid Closed-Loop system?
What do I need to know to be prepared for Omnipod 5?
If you are interested in the Omnipod 5 System, head over to our Getting Started page to order a free demo Pod!
Related Articles
Other topics
References and Disclaimers
*A separate prescription is required for the glucose sensor.
† Bolus for meals and corrections are still needed.
§ Fingerpricks required for diabetes treatment decisions if symptoms or expectations so not match readings.
‡The Pod has a waterproof IP28 rating for up to 7.6 metres for 60 minutes. The Controller is not waterproof. Please refer to the sensor manufacturer’s user guide for individual guidance.
1 ADA Standards of Medical Care in Diabetes – 2021. Diabetes Care. 2021;44(S1):S1-S232.
2 Study in 240 people with T1D aged 6 - 70 years involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Average overnight time (12AM-6AM) with high blood glucose in adults/adolescents and children for standard therapy vs Omnipod 5 = 32.1% vs. 20.7%; 42.2% vs. 20.7%. Average day time (6AM-12AM) with high blood glucose in adults/adolescents and children for standard therapy vs Omnipod 5 = 32.6% vs. 26.1%; 46.4% vs. 33.4%. Median overnight time (12AM-6AM) with low blood glucose in adults/adolescents and children for standard therapy vs Omnipod 5 = 2.07% vs. 0.82%; 0.78% vs. 0.78%. Median day time (6AM-12AM) with low blood glucose in adults/adolescents ad children for standard therapy vs Omnipod 5 = 1.91% vs. 1.08%; 1.17% vs. 1.62%. Average A1c in adults/adolescents and children, standard therapy vs. Omnipod 5 = 7.16% vs 6.78%; 7.67% vs 6.99%. Brown S. et al. Diabetes Care (2021).
3. Study in 80 people with T1D aged 2 - 5.9 yrs involving 2 weeks standard diabetes therapy followed by 3 months Omnipod 5 use in Automated Mode. Average time with high blood glucose overnight from CGM in standard therapy vs Omnipod 5 = 38.4% vs. 16.9%. Average time with high blood during daytime from CGM in standard therapy vs Omnipod 5 = 39.7% vs. 33.7%. Average time with low blood glucose overnight from CGM in standard therapy vs. Omnipod 5 = 3.41% vs. 2.13%. Average time with low blood glucose during daytime from CGM in standard therapy vs. Omnipod 5 = 3.44% vs. 2.57%. Average A1c in standard therapy vs. Omnipod 5 = 7.4% vs. 6.9%. Sherr JL, et al. Diabetes Care (2022)."
These modules are not a replacement for medical advice or training. Please always speak to a qualified healthcare professional about your options.
The information and other content provided in this article, or in any linked materials, are not intended and should not be construed as medical advice, nor is the information a substitute for professional medical expertise or treatment. If you or any other person has a medical question or concern, you should consult with your healthcare provider. Never disregard professional medical advice or delay in seeking it because of something that have read on this blog or in any linked materials. If you think you may have a medical emergency, call your doctor or emergency services immediately. The opinions and views expressed on this blog and website have no relation to those of any academic, hospital, health practice or other institution.