Omnipod® 5 showed strong clinical performance across a broad range of patient types1-3

TIME IN RANGE ACHIEVED IN THE REAL WORLD4,*
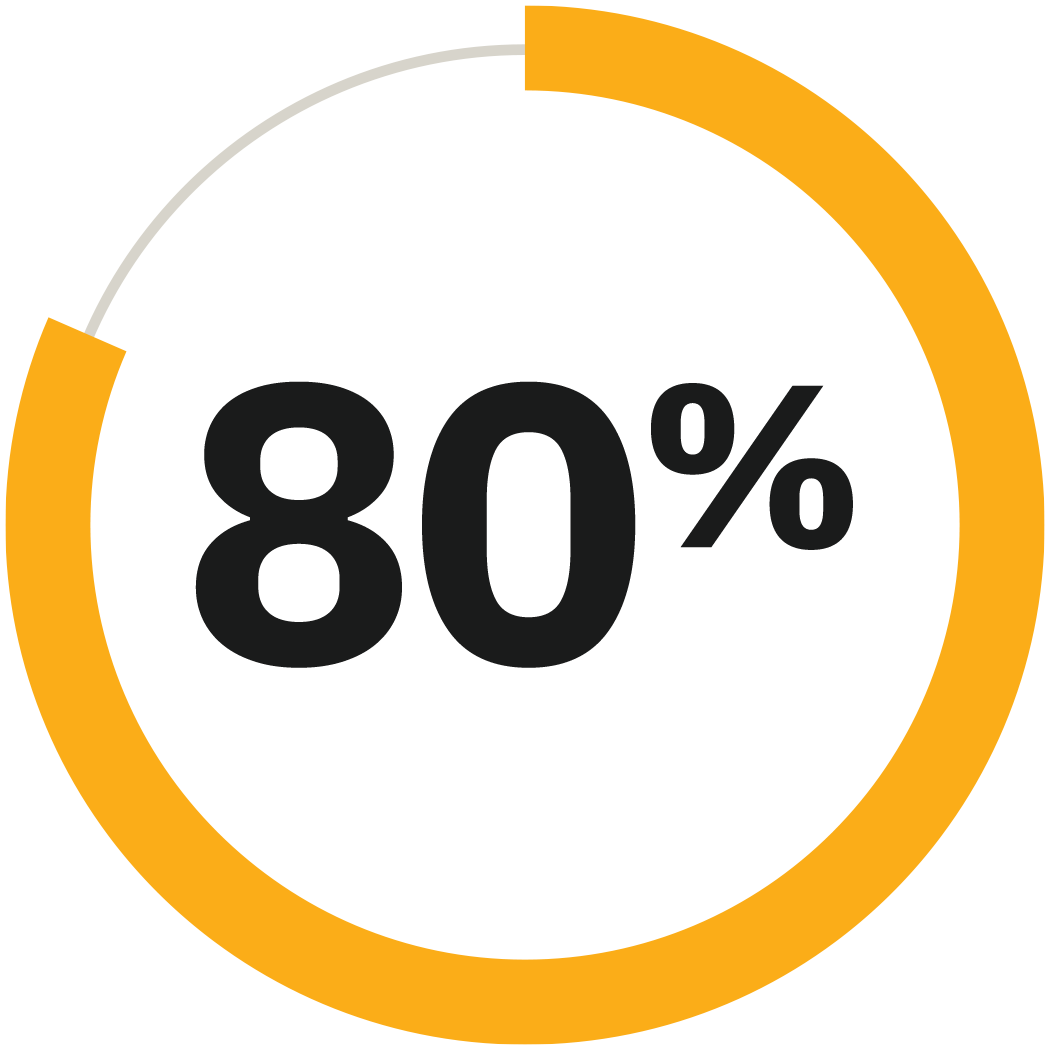
*Forlenza GP, DeSalvo DJ, Aleppo G, et al. Diabetes Technol Ther. 2024;26(8):514-525. Retrospective analysis of real-world evidence with 5,841 (20.4%) of the 28,612 adults with type 1 diabetes using Omnipod 5 who utilised the 110 mg/dL glucose target had 80% time in range or greater. Omnipod 5 results based on users with ≥90 days CGM data, ≥75% of days with ≥220 readings available.4
Many people living with T1D lose healthy years when they don’t have the right tools5
Glycaemic goals vs. the reality for patients on MDI
An HbA1c of <7% is an appropriate glycaemic goal for most people living with diabetes6
Average HbA1c for patients with T1D using MDI7
Omnipod 5 has shown to improve HbA1c and TIR while helping to protect against highs and lows8
MDI-user data
In the recent RADIANT randomised controlled trial1
- 189 participants (ages 4-70) with T1D were enrolled, 188 completed
- 58% paediatric (4.0–17.9 years)
- In France, Belgium, and the U.K.
- CGM: Abbott FreeStyle Libre 2
- All participants were previously on MDI therapy + Sensor
Omnipod 5 showed significant improvements compared to MDI therapy1
22.4%
MORE TIR
Omnipod 5 improved TIR by an average of 22% (compared to MDI therapy)
5.4
HOURS/DAY
Equal to an average of 5.4 hours more per day, with no increase in TBR
1% DECREASE IN HbA1c
51% of study participants with T1D with a baseline HbA1c ≥8% (64 mmol/mol) were able to improve their HbA1c by 1% compared to MDI therapy1
Pump-user data
Omnipod 5 improves results for those who have already tried a sensor and pump2
In the recent OP5-003 randomised controlled trial2
- 196 adults (age 18-70) with T1D in France and the U.S. were enrolled, 192 completed
- CGM: Dexcom G6
- All were on pump therapy (excluding AID) for ≥3 months
Omnipod 5 users achieved more TIR vs. prior pump therapy2
17.5%
MORE TIR
(p<0.0001)
4.2
HOURS/DAY
while decreasing TBR
Despite already using a non-AID insulin pump with CGM,
improvement in TIR and HbA1c was observed in patients who transitioned to Omnipod 5
Omnipod 5 lets users enjoy more of what matters most2
Omnipod 5 users found it easy to use9
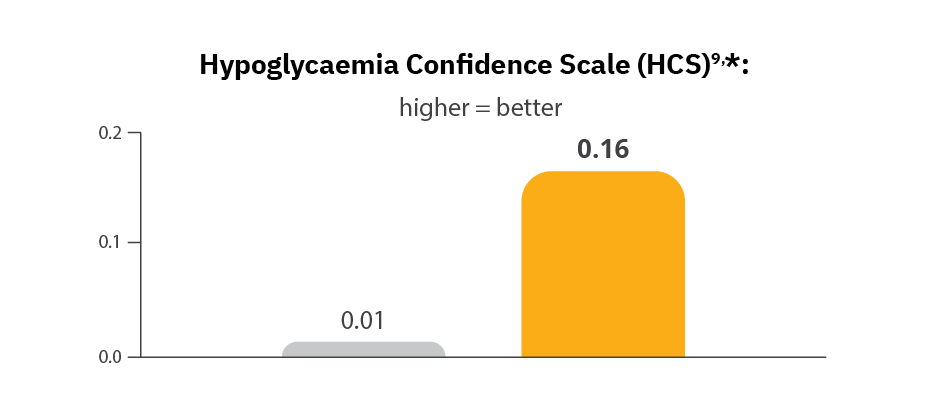
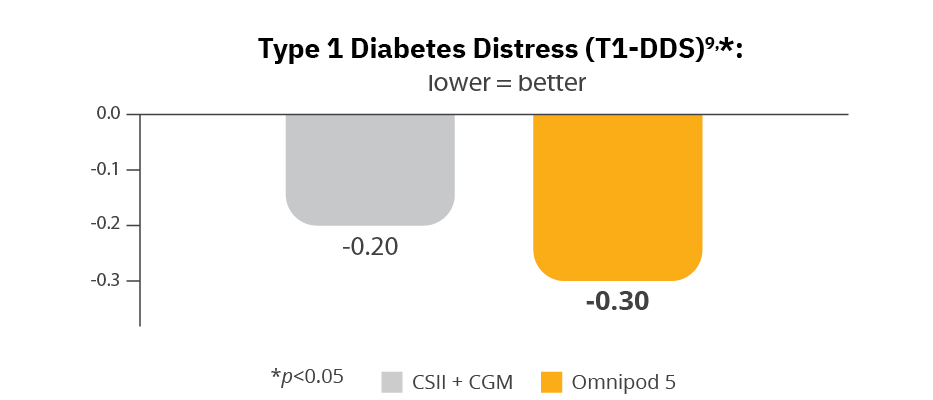
Omnipod 5 users reported more confidence in avoiding hypoglycaemia2,9
Omnipod 5 reduced diabetes distress2,9
"I love the freedom that the system gives me. It’s very simple and user friendly. It has undoubtedly changed my life for the better."
Joelle L.
Real Podder
Real-world data
Omnipod 5 users were successful in a real-world setting3
The Omnipod 5 real-world dataset consists of3:
- 69,902 U.S. Omnipod 5 users (ages ≥2) with ≥3 months of use
- Paediatric population (2–17 years): 22,162 (31.7%)
- 17.7 million person-days of Omnipod 5 use represented
- Previous therapy: Non-AID insulin pump: 67.1%, MDI: 19.1%, Unknown: 13.8%
- CGM: Dexcom G6
- Real-world evidence indicates effectiveness in non-study conditions and reaffirms findings from clinical trials
Use of the lowest Target Glucose (6.1 mmol/L [110 mg/dL]) was associated with a higher TIR3


>80% TIR achieved by 20% of adults at an average target of 6.1 mmol/L (110 mg/dL)4


In real world use, Omnipod 5 users experienced only 1.1% TBR at an average target glucose of 6.1 mmol/L (110 mg/dL)3
NO REASON TO DISCONNECT
Adults spent 94.1% of time in Automated Mode in a real-world setting3
With the tubeless, waterproof† Omnipod 5, there’s no need to disconnect during daily activities.‡


Omnipod 5 is effective even in candidates who may not be considered typical candidates for AID and are often overlooked3
Lowering target glucose significantly improved TIR10
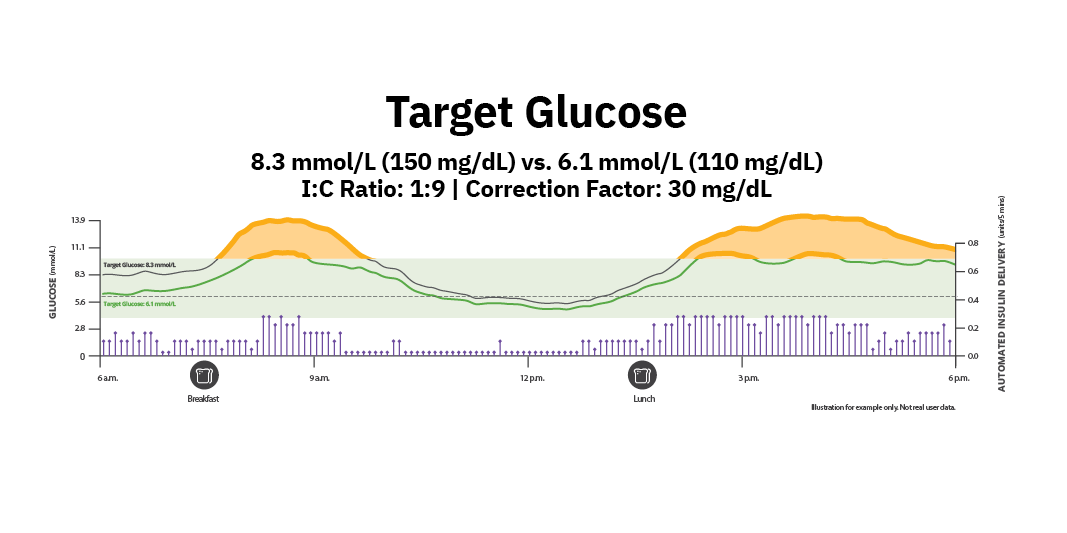

OPTIMISE RESULTS BY BALANCING AUTOMATED AND USER-INITIATED INSULIN
5 Target Glucose options for more flexibility
I:C ratio
Correction factor
Simplify mealtime factor bolusing with Custom Foods
More than 80% TIR with optimised settings in the real-world11
Get personalised support for Omnipod 5
†The Pod has an IP28 rating for up to 7.6 meters (25 feet) for 60 minutes. The Omnipod 5 Controller is not waterproof. Please consult sensor manufacturer user guide for sensor waterproof rating.
‡Device components including the Pod, Sensor, and Transmitter must be removed before Magnetic Resonance Imaging (MRI), Computed Tomography (CT) scan, or diathermy treatment. In addition, the Controller and smartphone should be placed outside of the procedure room. Exposure to MRI, CT, or diathermy treatment can damage the components.
AID, automated insulin delivery; CGM, continuous glucose monitor; HbA1c, glycated haemoglobin (A1C); I:C Ratio, insulin:carb ratio; MDI, multiple daily injections; TBR, time below range; TIR, time in range; T1D, type 1 diabetes.
References:
1. Wilmot E, et al. Presented at: ATTD; March 19–22, 2025; Amsterdam, NL. A 13-week randomized, parallel-group clinical trial conducted among 188 participants (age 4–70) with type 1 diabetes in France, Belgium, and the U.K., comparing the safety and effectiveness of the Omnipod 5 System versus multiple daily injections with CGM.
2. Renard E, et al. Diabetes Care. 2024. doi:10.2337/dc24-1550. A 13-week randomised controlled trial conducted among 194 adults (age 18-70 years) with type 1 diabetes in France and the U.S., comparing the safety and effectiveness of the Omnipod 5 Automated Insulin Delivery (AID) System versus conventional non-AID pump therapy and CGM (control). Mean time in range (3.9-10.0 mmol/L or 70-180mg/dL) at 13-weeks as measured by CGM: Control = 43.8%, Omnipod 5 = 61.2%. Mean adjusted difference Omnipod 5 vs. control = 17.5%, P<0.0001. Mean time in hypoglycaemic range (<3.9 mmol/L or <70mg/dL) at 13-weeks as measured by CGM: control =1.75%, Omnipod 5 = 1.18%. Mean adjusted difference Omnipod 5—control = -0.36%, P=0.005.
3. Forlenza GP, DeSalvo DJ, Aleppo G, et al. Diabetes Technol Ther. 2024;26(8):514-525. Retrospective analysis of real-world data from 6,525 Omnipod 5 users with type 1 diabetes at the Target Glucose of 6.1 mmol/L or 110 mg/dL who utilized MDI as prior therapy had a mean time in range of 70.8% and time below range of 0.96%.
4. Forlenza GP, DeSalvo DJ, Aleppo G, et al. Diabetes Technol Ther. 2024;26(8):514-525. Retrospective analysis of real-world evidence with 5,841 (20.4%) of the 28,612 adults with type 1 diabetes using Omnipod 5 who utilized the 110 mg/dL glucose target had 80% Time in Range or greater.
5. Type 1 Diabetes Index. https://www.t1dindex.org Accessed 1 Feb 2025.
6. Holt RIG, DeVries JH, Hess-Fischl A, et al. The management of type 1 diabetes in adults. A consensus report by the American Diabetes Association and the European Association for the Study of Diabetes (EASD). Diabetologia. 2021;64(12):2609-2652. doi:10.1007/s00125-021-05568-3.
7. Gandhi K, Ebekozien O, Noor N, et al. Insulin Pump Utilization in 2017–2021 for More Than 22,000 Children and Adults With Type 1 Diabetes: A Multicenter Observational Study. Clin Diabetes. 2024;42(1):56-64. doi:10.2337/cd23-0055
8. Brown S, et al. Diabetes Care. 2021;44:1630-1640. Prospective pivotal trial in 240 participants with T1D aged 6 - 70 yrs [adults/adolescents (n= 128; aged 14-70 yrs) children (n=112; aged 6-13.9 yrs)]. Study included a 14-day standard therapy (ST) phase followed by a 3-month Omnipod 5 hybrid closed-loop phase. Mean time >180 mg/dL in adults/adolescents and children, ST vs. 3-month Omnipod 5: 32.4% vs. 24.7%; 45.3% vs. 30.2%, P<0.0001, respectively. Mean time <70 mg/dL in adults/adolescents and children, ST vs. 3-month Omnipod 5: 2.9% vs. 1.3%, P<0.0001; 2.2% vs. 1.8%, P=0.8153, respectively. Results measured by CGM. Study funded by Insulet
9. Insulet Data on File OP5-003 Clinical Study Report 2024. A 13-week randomized, parallel-group clinical trial conducted among 194 adults (age 18–70) with type 1 diabetes comparing Omnipod 5 Automated Insulin Delivery System versus conventional pump therapy with CGM.
10. Forlenza G, et al. Presented at: ATTD; March 19–22, 2025; Amsterdam, NL. Real-world data from 403 people with type 1 diabetes aged ≥2 using the Omnipod 5 System who transitioned from the (150 mg/dL or 8.3 mmol/L) to (110 mg/dL or 6.1 mmol/L) Target Glucose. Each Target Glucose was used for a consecutive period of 14–90 days. Median time in Range (70–180 mg/dL) (3.9-10 mmol/L) improved 11.8% (p<0.05). Median time (<70 mg/dL or <3.9 mmol/L) +0.23% (p<0.05). Omnipod 5 results based on users with ≥75% of days with ≥220 readings available. Insulet Data on File. 05.15.25. RF-042025-00013.
11. Retrospective RWE data on file. 2025. Results shown for users with optimised settings including sufficient CGM data (≥75% of days with ≥220 readings), ≥90% time in Automated Mode, ≥5 bolus/day and an average Target Glucose of 110–115 mg/dL (6.1–6.4 mmol/L). Optimised settings: ISF × TDI ≤1500 (<= 83 mmol/L), I:C Ratio × TDI ≤350. RF-062025-00014.