Omnipod® 5

Omnipod 5 Automated Insulin Delivery (AID) System is the first wearable, tubeless, hybrid closed loop system that integrates with the Dexcom G6, Dexcom G7, ad Freestyle Libre 2 Plus sensors, for people with type 1 diabetes aged 2 years and older requiring insulin.
Omnipod 5 System: Three Simple Parts
Controller + Pod + CGM/Sensor
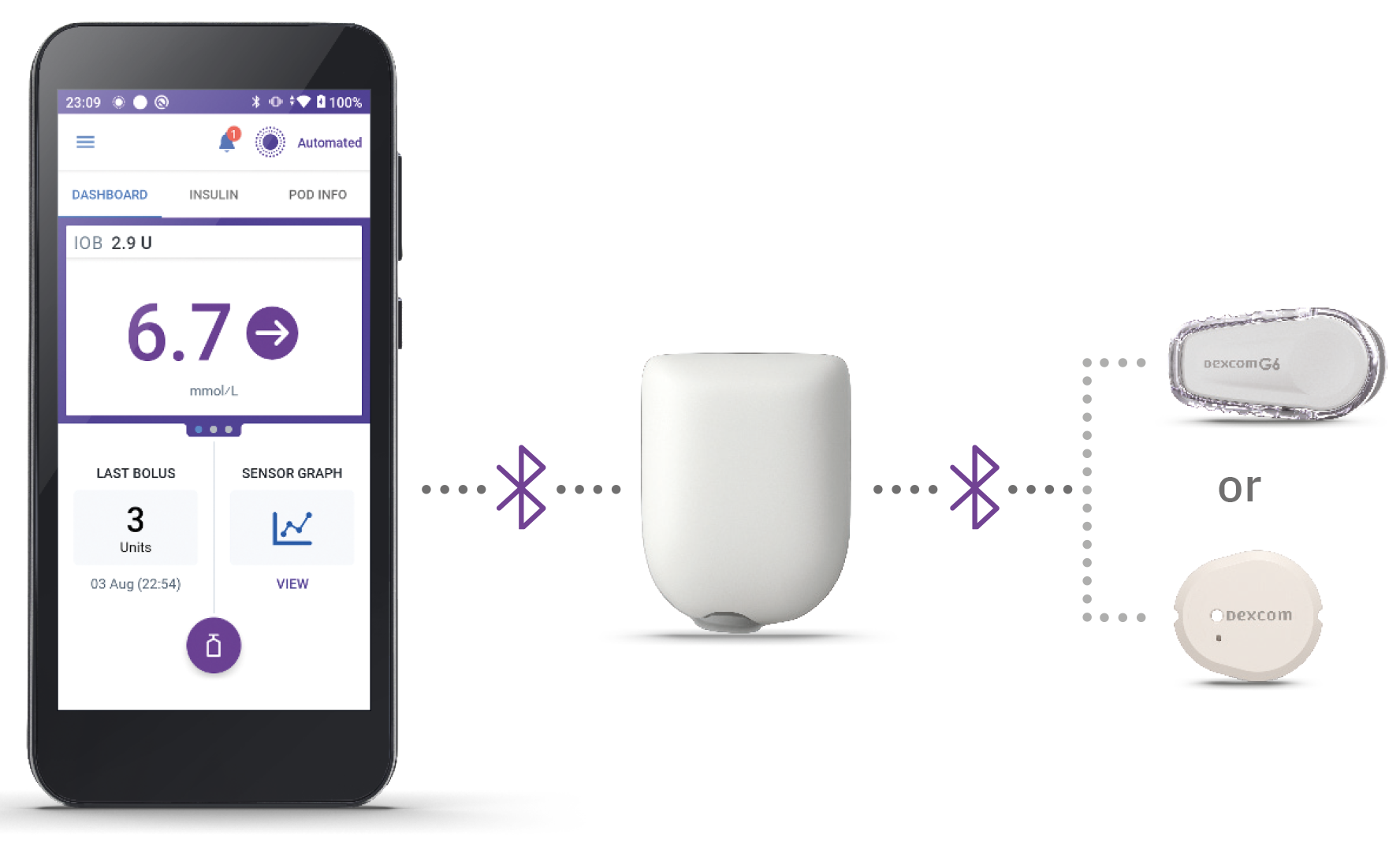

Omnipod 5 Controller
The Omnipod 5 Controller allows you to monitor and control the Pod using Bluetooth® wireless technology.



Pod
Tubeless, wearable, and waterproof,** the Pod, with built-in SmartAdjust™ technology, sits right on your patient’s body and automatically§ adjusts insulin delivery for up to 3 days or 72 hours.
§Automated Mode requires compatible CGM Sensor
**The Pod has an IP28 rating for up to 7.6 metres (25 feet) for up to 60 minutes. The Omnipod 5 Controller is not waterproof. Please consult sensor manufacturer user guide for sensor waterproof rating.
Sensor
CGM sensor continuously sends glucose values to the Pod, so your patients can get real-time data—without the finger pricks†.


Omnipod 5's Algorithm: SmartAdjust™ Technology
Predicts
Glucose 60 minutes into the future
Adjusts
Insulin delivery using the selected Target Glucose
Delivers
Insulin doses every 5 minutes (as needed)
Always adjusting, so you don't have to.
Omnipod® 5 with SmartAdjustTM technology automatically adjusts to your patients' personal needs by increasing, decreasing, or pausing insulin delivery every five minutes—which may help protect against highs and lows.1,2
Watch this video to learn more.
Improved Clinical Outcomes with Omnipod 5
Omnipod 5 improved glycaemic control in adults, adolescents and children with T1D in pivotal studies.1,2
76%
time in range (TIR) at a target of 6.1 mmol/L (110 mg/dL) in adults and adolescents (14–70 years) and 68% overall TIR in children (2- 13.9 years)1,2
HbA1c
was significantly reduced in very young children (2.0–5.9 years), children (6–13.9 years), adults and adolescents (14–70 years) by 0.5%, 0.7% and 0.4% (4.2, 6, and 7.8 mmol/mol) respectively1,2
33%
reduced time in hyperglycaemia in children and 24% in adults and adolescents1
60%
reduction in time in hypoglycaemia overnight and 46% overall in adults and adolescents1
Omnipod® 5 improved users’ quality of life in clinical studies3,4
- Parents of children using Omnipod 5 reported improvements in quality of life, sleep quality, confidence avoiding hypoglycaemia, and mental well-being3,4
- Adults using Omnipod reported reduction in diabetes distress and stress eating, confidence avoiding hypoglycaemia, and improved mental well-being3,4
In a 3-month clinical study, 3 cases of severe hypoglycaemia and 1 case of diabetic ketoacidosis (DKA) were reported in participants aged 6-70 years during Omnipod® 5 System use. These cases were not related to automated insulin delivery malfunction.1
References:
1. Brown S. et al. Diabetes Care. 2021;44:1630-1640. Prospective pivotal trial in 240 participants with T1D aged 6 - 70 yrs [Adults N=128, aged 14-70 years, Children N=112, aged 6-13.9 years). Study included a 14-day standard therapy (ST) phase followed by a 3-month Omnipod 5 hybrid closed-loop (HCL) phase. Mean time in hyperglycaemic range (>10.0 mmol/L or >180mg/dL) as measured by CGM in adults/adolescents and children ST vs. 3-mo Omnipod 5: 28.9% vs. 22.8%; 44.8% vs 29.7%, P<0.0001, respectively. Mean time in hypoglycaemic range (<3.9 mmol/L or <70 mg/dL) as measured by CGM in adults/adolescents and children ST vs. 3-mo Omnipod 5: 2.89% vs. 1.32%, P<0.0001; 2.21% vs. 1.78, P=0.8153, respectively. Mean time in range (3.9-10.0 mmol/L or 70-180mg/dL) in children as measured by CGM: ST = 52.5%, 3-mo Omnipod 5 = 68.0%, P<0.0001. Mean HbA1c: ST vs. Omnipod 5 use in adults/adolescents and children, respectively (7.16% vs 6.78% or 55 mmol/mol vs. 51 mmol/mol, P<0.0001; 7.67% vs 6.99% or 60mmol/mol vs 53 mmol/mol), P<0.0001). Average time above range (>10 mmol/L or >180 mg/dL) in children: ST vs. 3-month Omnipod 5 = 45.3% vs. 30.2%. Change is relative comparison. Average time above range (>10 mmol/L or >180 mg/dL) in adults: ST vs Omnipod 5: 32.4% vs. 24.7%. Change is relative. Mean time in hypoglycaemic range (<3.9 mmol/L or <70mg/dL as measured by CGM; 12AM - < 6AM) in adults as measured by CGM: ST: 2.07% vs. Omnipod 5: 0.82%, P<0.0001 Comparison is a relative change. Study funded by Insulet.
2. Sherr J. et al. Diabetes Care. 2022; 45:1907-1910. Single-arm multicenter clinical trial in 80 pre-school children (aged 2-5.9 yrs) with T1D. Study included a 14-day standard therapy (ST) phase followed by a 3-month AID phase with Omnipod 5 system. Mean time in hyperglycaemic range (>10.0 mmol/L or >180mg/dL) as measured by CGM in children ST vs. 3-mo Omnipod 5: 39.4% vs. 29.5%, P<0.0001, respectively. Mean time in hypoglycaemic range (<3.9 mmol/L or <70 mg/dL) as measured by CGM in children ST vs. 3-mo Omnipod 5: 3.43% vs. 2.46%, P=0.0204. Mean time in range (3.9-10.0 mmol/L or 70-180mg/dL) in very young children (2 - 5.9 yrs) as measured by CGM: ST = 57.2%, 3-mo Omnipod 5 = 68.1%, P<0.0001. Mean HbA1c: ST vs. Omnipod 5 use: 7.4% vs 6.9% or 57 mmol/mol vs 53 mmol/mol, P<0.05. Study funded by Insulet.
3. Hood KK et al. Diabetes Obes Metab. 2024. Study of 81 children aged 6-11.9 with type 1 diabetes using standard therapy (ST) for 2 weeks followed by 3-months of the Omnipod 5 System. Mean caregiver self-reported Hypoglycemia Confidence Scale score ST vs Omnipod 5: 3.34 vs 3.59, p-value <0.0001, respectively. During the Omnipod 5 pivotal trial, parents of children aged 6-11.9 years (N=82) experienced an improvement in emotional distress levels and mental well-being survey scores after 3 months of Omnipod 5 use compared to standard therapy: mean P-PAID-C score = 40.7 vs. 47.4; mean WHO-5 score = 72.9 vs. 67.5, respectively. During the Omnipod 5 pivotal trial, adults aged 18-70 (N=115) experienced an improved diabetes distress survey score after 3 months of AID use: mean: 1.48 vs. 1.64 (P < 0.001). During the Omnipod 5 pivotal trial, parents of children aged 6-11.9 years (N=82) experienced an improvement in emotional distress levels and mental well-being survey scores after 3 months of Omnipod 5 use compared to standard therapy: mean P-PAID-C score = 40.7 vs. 47.4; mean WHO-5 score = 72.9 vs. 67.5, respectively. During the Omnipod 5 pivotal trial, parents of children 6-11.9 years (N=82) experienced an improvement in sleep quality survey score after 3 months of Omnipod 5 use compared to standard therapy: mean PSQI Overall Sleep Quality Subscore = 0.70 vs 1.12, respectively. Study funded by Insulet.
4. Polonsky WH et al. Diabetes Res Clin Pract 2022;190:109998. Study of 115 adults aged 18-70 years with type 1 diabetes using standard therapy (ST) for 2 weeks followed by 3-months of the Omnipod 5 System. Mean self-reported Hypoglycemia Confidence Scale score ST vs Omnipod 5: 3.52 vs 3.65, p-value = 0.0002, respectively. Adults aged 18-70 (N=115) experienced reduction in eating distress score in T1-DDS survey after 3 months of Omnipod 5 use vs baseline: 1.73 vs. 1.97, (P=0.0003), respectively. Study funded by Insulet.
Initiating a Patient on Omnipod 5
Help your patients get a successful start on Omnipod® 5.
Omnipod 5 Simplifies Data Management
With Glooko®, your patient can access all of their diabetes information in one easy-to-use platform and easily share it with you.
Omnipod 5 Resources for your Patients
All of the resources, all in one place. Visit our resources page for User Guides, How-to-Videos, FAQs and more.